Figures & data
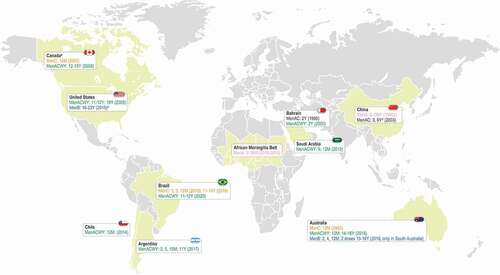
Figure 2. Evolution over time of meningococcal vaccination strategies in European countries.
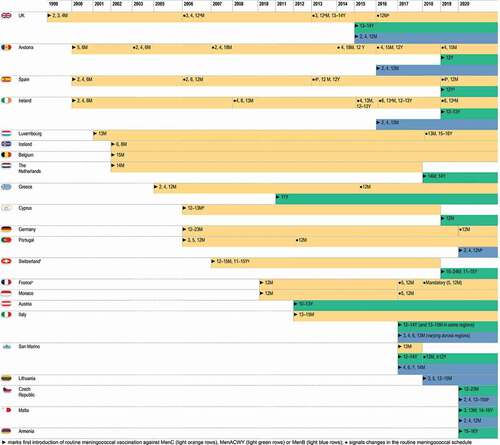
Figure 3. Evolution of serogroup-specific invasive meningococcal disease incidence and meningococcal vaccination strategies for 5 European countries.

Figure 4. Distribution of reported serogroup-specific invasive meningococcal disease cases by age in European countries, 2014–2018
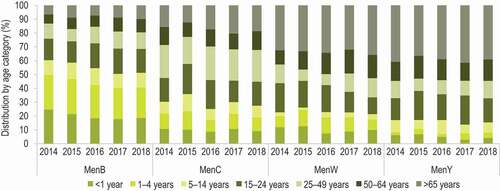
Figure 5. Introduction of routine meningococcal vaccination and current vaccination schedule in non-European countries