Figures & data
Figure 1 Potential mechanism of action of ozanimod in inflammatory bowel disease. Binding of ozanimod induces S1P1 receptor internalization, blocking the capacity of lymphocytes to egress from lymphoid tissue into circulation.Citation5,Citation7,Citation8 The mechanism by which ozanimod exerts therapeutic effects in inflammatory bowel disease is unknown but may involve the reduction of lymphocyte migration into, or direct effects on, the gut.Citation5 Although S1P is prominent throughout the bloodstream, it is not depicted in the bloodstream on the left for simplicity in this conceptual illustration. aIncluding T cells and B cells. bInnate immune cells, which are responsible for antigen presentation and immunosurveillance, include macrophages, monocytes, and natural killer cells, among others. Adapted from Sands BE, Schreiber S, Blumenstein I, Chiorean MV, Ungaro RC, Rubin DT. Clinician’s guide to using ozanimod for the treatment of ulcerative colitis. J Crohns Colitis. 2023;jjad112. Creative Commons.Citation9
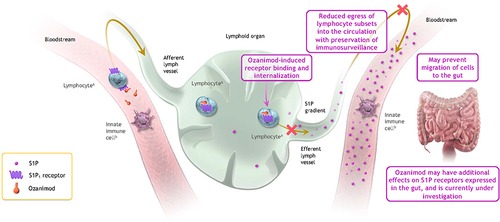
Table 1 Adverse Events in Patients with UC in the Phase 3 True North studyCitation15
Table 2 Incidence of Adverse Events of Special Interest in Patients with UC and MS Treated with Ozanimod 0.92 mgCitation6,Citation15
Figure 2 Mean ALC over time in the induction and maintenance periods of the True North study in patients with UC.Citation24 Patients who received continuous placebo (placebo) had stable levels of mean ALC over time, remaining within the normal range (1.02–3.36 × 109/L). Patients who continuously received ozanimod during the induction and maintenance periods (ozanimod/ozanimod) had sustained ALC reductions over time, which began to recover within 8 weeks in patients who discontinued ozanimod during the maintenance period (ozanimod/placebo). Error bars denote standard error. Reprinted from Sands BE, Schreiber S, Blumenstein I, Chiorean MV, Ungaro RC, Rubin DT. Clinician’s guide to using ozanimod for the treatment of ulcerative colitis. J Crohns Colitis. 2023;jjad112. Creative Commons.Citation9
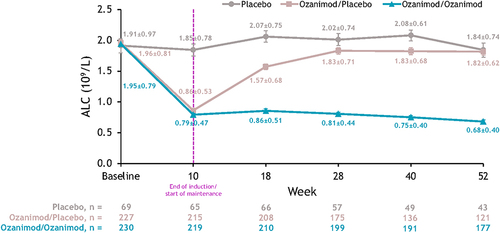
Table 3 Checklist for Ozanimod Treatment in Patients with UC
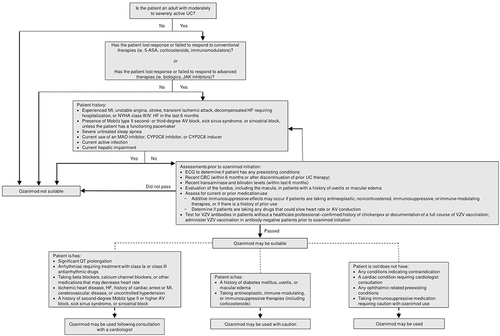
Figure 3 Guide to establishing suitability for ozanimod treatment in patients with UC.Citation6 Ozanimod may be suitable for adult patients with moderately to severely active UC. The ideal patient has lost response or failed to respond to conventional or advanced therapies. Patients must pass preinitiation assessments as described in the US prescribing information. Consultation with a cardiologist is recommended prior to treatment initiation in patients with preexisting cardiac conditions. Caution is advised for patients at risk for macular edema and those taking immunosuppressive drugs. The prescribing information also outlines patients for whom ozanimod is contraindicated or not recommended. This figure outlines the key issues but is not comprehensive of all considerations detailed in the prescribing information.