 ?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.
?Mathematical formulae have been encoded as MathML and are displayed in this HTML version using MathJax in order to improve their display. Uncheck the box to turn MathJax off. This feature requires Javascript. Click on a formula to zoom.Abstract
Context
Musculoskeletal system disorders (MSD) are prevalent around the world affecting the health of people, especially farmers who work hard in the field. Karen farmers use many medicinal plants to treat MSD.
Objective
This study collects traditional plant-based remedies used by the Skaw Karen to treat MSD and evaluates their active phytochemical compounds.
Materials and methods
The ethnobotanical study was conducted in six Karen villages in Chiang Mai province using semi-structured interviews were of 120 informants. The data were analyzed using ethnobotanical indices including use values (UV), choice value (CV), and informant consensus factor (ICF). Consequently, the 20 most important species, according to the indices, were selected for phytochemical analysis using LC–MS/MS.
Results
A total of 3731 use reports were obtained for 139 species used in MSD treatment. The most common ailments treated with those plants were muscular pain. A total of 172 high-potential active compounds for MSD treatment were identified. Most of them were flavonoids, terpenoids, alkaloids, and steroids. The prevalent phytochemical compounds related to treat MSD were 9-hydroxycalabaxanthone, dihydrovaltrate, morroniside, isoacteoside, lithocholic acid, pomiferin, cucurbitacin E, leonuriside A, liriodendrin, and physalin E. Sambucus javanica Reinw. ex Blume (Adoxaceae), Betula alnoides Buch.-Ham. ex D.Don (Betulaceae), Blumea balsamifera (L.) DC. (Asteraceae), Plantago major L. (Plantaginaceae) and Flacourtia jangomas (Lour.) Raeusch. (Salicaceae) all had high ethnobotanical index values and many active compounds.
Discussion and conclusions
This study provides valuable information, demonstrating low-cost medicine plants that are locally available. It is a choice of treatment for people living in remote areas.
Introduction
Thailand is blessed with a rich biodiversity. More than 30% of the country or about 16 million hectares are covered by natural forests (Royal Forest Department [RFD] Citation2022). These forests are a source of countless plants, many of which are used by ethnic people of Thailand. The knowledge about plant usage is passed from generation to generation and has developed into unique traditional knowledge of each ethnic group (Bruchac Citation2014). Traditional medicine has played an important role for the subsistence and health of ethnic people, especially for those who live in remote areas (Cadena-González et al. Citation2013; Junsongduang et al. Citation2020). In Thailand, ethnic people have used medicinal plants for treating many disorders including digestive system disorders, infections/infestations, nutritional disorders, and musculoskeletal system disorders (Pongamornkul Citation2017; Phumthum et al. Citation2018; Kantasrila et al. Citation2020). There are more than 2000 species of vascular plants used to treat ailments in daily life of ethnic people (Phumthum et al. Citation2018). More than 30% of these species were reported to be related to musculoskeletal system disorders (MSD) (Phumthum et al. Citation2018).
MSD are non-communicable diseases causing short-term and long-term pains (Woolf and Pfleger Citation2003; Collins and O’Sullivan 2010). Approximately 2 billion people around the world suffer from MSD, such as sprains, strains, back pain, carpal tunnel syndrome, osteoarthritis, lower back pain, upper limb disorders, sprains, fractures, dislocations, and connective tissue disease (Cieza et al. Citation2020). MSD are a main problem leading to disability and death (Hignett and Fray Citation2010). MSD are influenced by many factors, such as age, lifestyle, physical activity, and career (Hignett and Fray Citation2010; Sombatsawat et al. Citation2019; World Health Organization [WHO] Citation2019). In this context, agriculture is a risk occupation, which is one of the most important causes of MSD due to the hard work, lifting, and carrying heavy loads, working with farm vehicles, and a high risk of accidents (Kang et al. Citation2016; Sombatsawat et al. Citation2019). Farmers from various countries, such as India (Gupta and Tarique Citation2013), Iran (Omran et al. Citation2015), Thailand (Puntumetakul et al. Citation2011; Sombatsawat et al. Citation2019), Pakistan (Saeed et al. Citation2021) and Korea (Kang et al. Citation2016) suffer from MSD. The most prevalent symptoms, which occur among these farmers are pain in various body parts, especially in the lower back, neck, and shoulders. These problems have limited the working ability, causing decrease of farm income (Oh et al. Citation2011; Hartvigsen et al. Citation2018). Farmers, which are a majority occupation in Thailand, are affected by MSD like the farmers around the world (Puntumetakul et al. Citation2011; Sombatsawat et al. Citation2019; Kantasrila et al. Citation2020). People around the world have used both traditional medicine and modern medicine to cure MSD. Traditional medicine has experienced growing popularity (Sheng-Ji Citation2001), especially for treating muscular pain, rheumatism, fractured bones, etc. Many active phytochemical compounds such as flavonoids, terpenoids, alkaloids in medicinal plants possess the potentiality to prevent the generation of free radicals and inflammation, which are two highly important causes of MSD (Kanadaswami et al. Citation2005; Fusco et al. Citation2007; Barros et al. Citation2011; Carocho and Ferreira Citation2013). Medicinal plants are one of the best alternative therapies for treating MSD in communities of tribes and other parts of the world (Cameron and Chrubasik Citation2013).
Thailand has a high diversity of ethnic groups such as Karen, Kui, Mon, Hmong, Lahu, Akha, etc. (Schliesinger Citation2000). There are more than thirty ethnic groups and together they count about 1.1 million people who live along the borders of Thailand (Department of Social Development and Welfare [DSDW] Citation2016). Sgaw Karen is the largest of the ethnic groups in Thailand with more than 500,000 people in Tak, Mae Hong Son, Chiang Mai, Ratchaburi, and Kanchanaburi provinces (DSDW Citation2016). Their subsistence mostly depends on traditional agriculture, especially rice cultivation in swidden fallows (Schliesinger Citation2000; Santasombat Citation2004). Their agricultural work relies primarily on manual labor because of lack of agricultural machines. This causes various health problems associated with uncomfortable postures such as bending, twining, lifting, and carrying heavy loads (Luangwilai et al. Citation2014; Sombatsawat et al. Citation2019). These activities are the causes of MSD. Many medicinal plants are used to treat MSD among the Karen people because healthcare centers are limited and often located far away. Moreover, modern medicine, which are used to treat MSD, are costly and have a lot of side effects. Even if medicinal plants are very important to Karen people for treating MSD, studies of such medicinal plants are limited (Kantasrila et al. Citation2020). The study about medicinal plants involves alternative therapies, which are one of the good choices to treat MSD among Karen communities due to medicinal plants being available and easy to get. Therefore, in this study, we aimed to answer the following questions: 1) What were the most prevalent phytochemical compounds in medicinal plants related to treatment of MSD? 2) Which species had the highest number of phytochemical compounds related to MSD determined using LC-MS/MS method? 3) Can these ethnomedicinal species with high ethnobotanical index scores be scientifically validated based on the results of phytochemical analysis? This study is a comprehensive review survey of phytochemical constituents of medicinal plants to screen important medicinal plant species for further drug development and produce new product of medicine form plants. In addition, this study highlights the importance of the usage medicinal plants to treat MSD among ethnic groups and provides significant information to preserve traditional knowledge about medicinal plants usage for future generations.
Materials and methods
Study site
The ethnobotanical study was conducted in Chiang Mai Province, northern Thailand, which is covered by high mountains and forested area (Asavachichan Citation2010). Chiang Mai is one of the provinces with large Skaw Karen population (DSDW Citation2016). There are more than 33,000 households, and approximately 30% of Skaw Karen population in Thailand, live in Chiang Mai (DSDW Citation2016). Field investigations were conducted in six Karen villages from five districts in Chiang Mai province, Thailand (). Basic information of the villages is shown in .
Figure 1. Locations of the five sub-districts in Chiang Mai province where ethnobotanical knowledge was studied in six villages.
Table 1. Basic information of the six studied Karen villages in Chiang Mai, Thailand.
Ethnobotanical study
The field research was conducted from 2017–2020. Initially, the chief of each village was contacted and informed about the aims of the study. Then, the key informants, including folk healers and those who were experienced in using traditional medicine in each village, were selected by snowball sampling (Espinosa et al. Citation2012). Each key informant was interviewed about medicinal plants that they used for treating musculoskeletal disorders (MSD) using semi-structured interviews (Ethical approval: the Research Ethics Committee of Chiang Mai University, protocol code CMUREC 63/029 9 July 2020). The key questions included local name, plant part used, mode of preparation, mode of administration, frequency of usage, dosage of medication, plant habitats, illnesses treated with their symptomatology and contraindications to medicinal plants usage. The surveys were conducted in home gardens, agricultural areas, and forests around the villages.
Plant samples were collected for identification, at least three specimens were collected from each plant mentioned by the key informants for identification. Plant materials were dried and identified to their species name using Flora of Thailand and related literature. The current scientific names and family names were verified based on The Plant List (www.theplantlist.org). Voucher specimens were deposited at the QBG herbarium.
Quantitative ethnobotanical surveys were conducted in each village with 20 informants, including equal number of men and women, with ages ranging from 20–70 years, who were selected by snowball sampling. Each informant was independently interviewed about plants used to treat MSD by showing photos and plant samples covering parts used, methods of preparation, and route of administration. The interviews were done in local language with a local interpreter.
Ethnobotanical data analysis
The medicinal plants were classified to groups by plant part used (roots, leaves, stems, etc.), mode of preparation (decoction, burned, ground, etc.), and route of administration (oral ingestion, liniment, etc.). The illnesses were grouped into categories of MSD following the International Classification of Primary Care, Second edition (ICPC-2) (Wonca International Classification Committee [WICC] Citation2005).
The information of medicinal plants was transcribed into use-reports (UR) following illnesses of the MSD. Each “use-report” refers to the use of a particular species to treat a specific ailment in an MSD category in a Karen village. The use-reports were the basis for calculating ethnobotanical indices including Use Value (UV), Choice Values (CV) and Informant Consensus Factor (ICF).
Use value (UV)
UV was proposed by Prance et al. (Citation1987) to evaluate the importance of plant species.
where Ui represents the number of use reports for each species, which is cited by informants and N is the total number of informants. The UVs are high for plants with many use-reports indicating that the plant is important and useful (Khuankaew et al. Citation2014; Samoisy and Mahomoodally Citation2016).
Choice values (CV)
CV was proposed by Kremen et al. (Citation1998) to define the most preferred species in each use category. In this study, CV was used to measure the relative preference for treatment of MSD. It was calculated as:
where Pcs is percentage of informants who cite certain species for MSD.
Sc is total number of species mentioned for treatment of MSD by all informants.
CV ranges from 0 to 100, with a higher value indicating these species were strongly preferred by the informants.
Informant consensus factor (ICF)
ICF was used to test the degree of homogeneity among informants for medicinal plants used in each category (Trotter and Logan Citation1986).
where Nur refers to the number of use-reports for a particular ailment category and Nt refers to the number of plants species recorded in that same category. ICF ranges from zero to one. When it is low (near 0), it implies that most informants report different plants for a particular ailment category. Therefore, the informants had completely different knowledge to treat specific illness, and there has been no exchange of medicinal plants knowledge among informants. On the other hand, the ICF value is high (approaching 1) when there are a few plants used in a particular ailment category and it implies that the knowledge has been exchanged among the informants (Heinrich et al. Citation1998).
Phytochemical analysis
Plant materials and extraction
Twenty medicinal plants that had high ethnobotanical index scores and were present in most studied villages were selected for further phytochemical analysis. Only the parts of the plants mentioned by the key informants were harvested. The samples were cleaned with tap water and dried in a hot air oven at 60 °C for 24 h. Then, the dried samples were ground by electric blender into powder. Dried sample (5 g) was extracted with 8 mL of distilled water using sonication for 30 min. The solution was filtered through 0.45 µm diameter, 25 mm Nylon membrane syringe filters and kept in amber glass bottles at −20 °C.
Phytochemical study; Liquid Chromatography with Tandem Mass Spectrometry (LC–MS/MS)
The methods and condition of LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry) in this study follow Panya et al. (Citation2020). Each sample solution was dissolved in DMSO in the concentration of 5000 ppm for further analysis with LC-MS/MS (Agilent 6545 LC/Q-TOF, Agilent, CA, USA) using Porshell 120 EC-C18, 2.7 µm, 2.1 × 100 mm C18 column, with gradient system of two mobile phases. The mobile phase A comprised of water and 0.1% acetic acid and mobile phase B comprised of acetonitrile (CAN) and 0.1% acetic acid for 60 min. The gradient elution program was set as follows: 100% A (0–30 min), 50% A (30–40 min), 0% A (40–51 min), 50% A (51–60 min). This was followed by a 10 min equilibrium period to the injection of the next sample. The injection volume of each sample was 3 µL, flow rate 0.2 mL/min. The UV spectra were recorded between 210 and 400 nm. The MS analysis condition of each HPLC peak included in the MS positive mode, Dual AJS ESI ion source, with the following operating parameters: sheath gas temperature 350 °C, sheath gas flow 11 L/min, gas temperature 320 °C, drying gas 8 L/min, nebulizer pressure 35 psi. The identified phytochemical compounds from LC-MS/MS methods were searched for their pharmaceutical activities from previous publications focused on anti-inflammatory, antioxidative and analgesic properties related to MSD. The selected compounds with MSD properties were further analyzed for the most prevalent compounds appearing in most medicinal plants.
Results
Diversity of medicinal plants used to treat MSD
From the ethnobotanical study, a total of 3731 use reports from 139 species (117 genera, 62 families) were recorded for treating MSD by the Karen people (). Most of them (135 species) were angiosperm. The most dominant families of MSD medicinal plants were Fabaceae (10 species, 233 use reports), Asteraceae (8 species, 457 use reports), Zingiberaceae (8 species, 231 use reports), Poaceae (6 species, 164 use reports), and Rubiaceae (6 species, 19 use reports), respectively ().
Table 2. Medicinal plants used to treat musculoskeletal system disorders (MSD) in the six Karen villages in Chiang Mai, Thailand.
Table 3. The plant families, number of species, and use reports of the medicinal plants used to treat MSD among the Karen in the six villages in Chiang Mai province, Thailand.
The 3731 use reports belonged to 23 ailment categories in the ICPC-2 classification (WICC, Citation2005). The categories with high numbers of use reports included muscle pain (33%), followed by flank/axilla symptom/complaint (23%), and back symptom/complaint (12%) (). On the other hand, there were many categories, which had small numbers of use report and species, e.g., sprain/strain of knee, chest symptom/complaint, hand/finger symptom/complaint and neck symptom/complain categories. These categories had only one species each for treating MSD. Moreover, there were five species, which were used to treat the same ailment and they were prepared in the same way in all six villages, including Betula alnoides, Blumea balsamifera, Chromolaena odorata (L.) R.M.King & H.Rob. (Asteraceae), Sambucus javanica, and Sambucus simpsonii Rehder (Adoxaceae).
Table 4. The ICF score for each ailment category (ordered by ICF value).
Ethnobotanical indices: use values, choice values and informant consensus factor
Use values (UV)
The use value (UV) index was calculated to find the most important medicinal plants, which were used to treat MSD among the Skaw Karen people based on number of use reports. The UV ranged from 0.01–1.49 (). Ten species with high UV included Sambucus javanica (1.49), Betula alnoides (1.15), Blumea balsamifera (1.13), Plantago major (1.10), Crinum asiaticum L. (Amaryllidaceae) (1.02), Pothos chinensis (Raf.) Merr. (Araceae) (1.02), Flacourtia jangomas (0.96), Pothos scandens L. (Araceae) (0.93), Cymbopogon citratus (DC.) Stapf (Poaceae) (0.86), and Sambucus simpsonii (0.84). There were 12 species with low UV (=0.01) with only a single use report for each of them, e.g., Clausena excavata Burm.f. (Rutaceae), Clematis smilacifolia Wall. (Ranunculaceae), Dendrocalamus brandisii (Munro) Kurz (Poaceae), and Diplazium esculentum (Retz.) Sw. (Athyriaceae).
Choice values (CV)
The Choice value (CV) was used to find the most preferred species for treating MSD based on number of informants. In this study, CV ranged from 0.01–0.56 (). The species with high CV values were the same as the species with high UV values, e.g., Sambucus javanica (0.56), Betula alnoides (0.51), Blumea balsamifera (0.48), Plantago major (0.43), Crinum asiaticum (0.40), Pothos chinensis (0.40), Flacourtia jangomas (0.40), Pothos scandens (0.35), Centella asiatica (L.) Urb. (Apiaceae) (0.34). On the other hand, there were 33 species, which had low CV values. These species had only one or two informants who mentioned their use.
Informant consensus factor (ICF)
The ICF was calculated to find any agreement between informants concerning plants used in each category. In this study, ICF ranged from 0.00–1.00 (). The ailment category with the highest ICF values was [sprain/strain of knee (1.00), pain (0.90), followed by flank/axilla symptom/complaint (0.88), back symptom/complaint (0.85), fracture: other (0.87) and sprain/strain of joint NOS (0.87)] category. However, there were five categories with the ICF values equal to zero, including foot/toe symptom/complaint, sprain/strain of ankle, chest symptom/complaint, hand/finger symptom/complaint and neck symptom/complaint.
Phytochemical compounds in important medicinal plants related to MSD
Twenty species, which had high UV and CV values, were selected for phytochemical analysis. Among these, two species including Sambucus javanica and S. simpsonii were first reported for their phytochemical compound and pharmacological activity. The chemical compositions of the methanol extracts of 20 promising plant species were analyzed using LC-MS/MS and 1674 compounds were reported. Among these, 172 compounds have been recorded for their ability to treat MSD and related diseases such as anti-arthritic, analgesic, anti-inflammatory, anesthetic, antinociceptive, and antioxidative properties (). The main class of secondary metabolites which had high possibility to be active components were flavonoids (45 compounds), terpenoids (42 compounds), alkaloids (29 compounds) and steroids (14 compounds) (). The most prevalent active compounds, which have phytochemistry properties related to MSD were 9-hydroxycalabaxanthone, dihydrovaltrate and morroniside (). These compounds were found in 11 species. 9-Hydroxycalabaxanthone (C24H24O6) is a member of xanthones, dihydrovaltrate (C22H32O8) is a member of iridoid monoterpenoids and morroniside (C17H26O11) is a member of iridoid monoterpene glycosides. Other most prevalent compounds were isoacteoside (C29H36O15) and lithocholic acid (C24H40O3). These active compounds were found in 9 species. Pomiferin (C25H24O6) was found in 8 species. Cucurbitacin E (C32H44O8), leonuriside A (C14H20O9), liriodendrin (C34H46O18), and physalin E (C28H32O11) were found in 7 species. Eurycoma longifolia Jack (Simaroubaceae) and Flacourtia jangomas had the highest number of active compounds (29 compounds) followed by Croton kongensis Gagnep. (Euphorbiaceae) (28 compounds), Plantago major (27 compounds), Blumea balsamifera (27 compounds), Betula alnoides (27 compounds), and Tadehagi triquetrum (L.) H.Ohashi (Fabaceae) (27 compounds) ().
Figure 2. The proportion of number of active compounds in each group which related to MSD treatments found in 20 selected medicinal plant species.
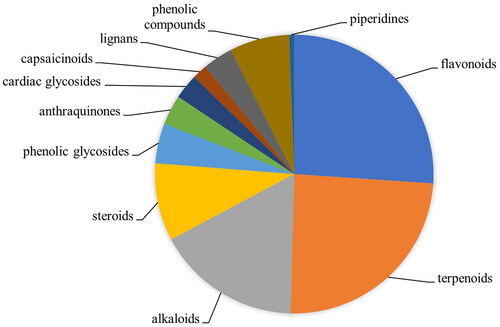
Figure 3. The numbers of active compounds related to MSD treatments found in 20 selected medicinal plant species.
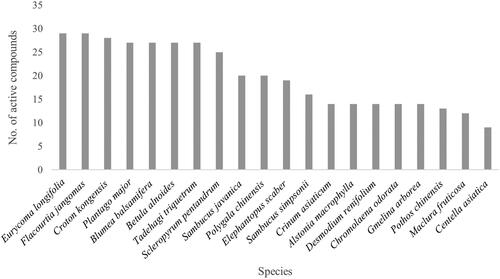
Table 5. The 172 active compounds found in the 20 medicinal plant extracts using for MSD treatments in the six Karen villages in Chiang Mai province, Thailand.
Discussion
Diversity of medicinal plants used to treat MSD
Karen people use a diversity of plants to treat MSD. The species number ranged between 46–94 species in the studied villages. In total, 139 species were recorded which correspond to 80% of species which were reported to treat MSD among Karen people in previous study (Kantasrila et al. Citation2020) and they made up 20% of medicinal plant species in Thailand reported by Phumthum et al. (Citation2020). High number of species could be related to high prevalence of MSD among the Karen people. Most Karen are farmers with rural livelihoods that may provoke symptoms and illnesses related to MSD (Phumthum et al. Citation2020).
Important species for treating MSD
The use value (UV) and choice values (CV) made it possible to evaluate important species for treating MSD. Plants with high UV tend to have good properties for treating ailments (Mahmood et al. Citation2012; Malik et al. Citation2018) while CV determined the most preferred plant species used in each ailment category (Kremen et al. Citation1998). Both the UV and CV indices are useful for selecting plants for further phytochemical and pharmacological studies (Malik et al. Citation2018; Nguyen et al. Citation2020b). The species with the highest UV and CV was Sambucus javanica. In this study, it was used in 11 categories of the classification of MSD such as ankle symptom/complaint, back symptom/complaint, fracture: other, hand/finger symptom/complaint, etc. The use of this plant was reported from all the studied villages and by most informants (80%). This plant is well-known for its medicinal properties to treat MSD, not only by the Karen, but several other ethnic groups, including Akha (Anderson Citation1993; Inta Citation2008), Hmong (Srithi Citation2012), Lua (Srithi Citation2012), Mien (Anderson Citation1993; Srithi Citation2012), and Tai Yuan (Srithi Citation2012).
Sambucus simpsonii, is another species with high UV and CV similar to those of S. javanica. These two species were considered to be the same species (ethno-species) by the Karen people since both of them were called by the same name. Other species with high UV and CV values included Betula alnoides, Blumea balsamifera, Plantago major, Crinum asiaticum, Pothos chinensis, Flacourtia jangomas, Pothos scandens, Polygala chinensis L. (Polygalaceae), Cymbopogon citratus, Duhaldea cappa, Curcuma longa L. (Zingiberaceae), Elephantopus scaber, and Gmelina arborea Roxb. (Lamiaceae). These plants are commonly used as medicine to treat MSD in Thailand and around the world (). For example, B. alnoides has been reported for its properties to treat muscle pain (Inta and Pongamornkul Citation2015b), back pain (Boonkorn Citation1997; Archam Citation2011), bone pain (Boonkorn Citation1997; Archam Citation2011), and joint pain (Muangyen Citation2013). In India, it has been used for treating microfractures, dislocated bones, joint pains, and sprain (Manandhar Citation1995; Singh et al. Citation1996; Changkija Citation1999).
Table 6. Medicinal plant used to treat musculoskeletal system disorders (MSD) among the Skaw Karen in Thailand.
Blumea balsamifera has also been reported for its uses to treat rheumatism and lumbago in many south-east Asia countries (Pang et al. Citation2014a). This plant has a strong aromatic fragrance from its many phytochemical constituents (Shirota et al. Citation2011). The important active compounds, such as sesquiterpenoids and flavonoids, are useful for their anti-inflammatory and antioxidative activities (Nessa et al. Citation2004; Shirota et al. Citation2011; Pang et al. Citation2014b).
Plantago major is one of the most abundant species, which could be found in many areas, especially in home gardens. Therefore, it is always available for use at any time and in this study, it was used in 12 MSD ailment categories. The species was also commonly used for treating MSD among other ethnic groups in Thailand. For example, Tai Yai (Areekun and Onlamun Citation1978; Udompanid Citation2012), Mien (Areekun and Onlamun Citation1978; Anderson Citation1993), Akha (Anderson Citation1993), and Hmong (Anderson Citation1993). It was used to treat rheumatic ailments, bone fractures, and muscle pains. Moreover, it has many active compounds like flavonoids, terpenoids, pectins, iridoid glycosides, and tannins which are all related to anti-inflammatory and antioxidant activities (de Padua 1999; Hussan et al. Citation2015).
In addition, Crinum asiaticum (Mahomoodally et al. Citation2021), F. jangomas (Sasi et al. Citation2018), Cymbopogon citratus (Kumar et al. Citation2010b), Duhaldea cappa (Kaur et al. Citation2017), Curcuma longa (Ahmad et al. Citation2010; Oghenejobo and Bethel Citation2017), Elephantopus scaber (Wang et al. Citation2013; Wang et al. Citation2014) and Gmelina arborea (Kaswala et al. Citation2012; Lawal Citation2016; Arora and Tamrakar Citation2017; Gandigawad et al. Citation2018) all have many active compounds that determine their medicinal properties, based on antioxidative, analgesic, antinociceptive, antipyretic and anti-inflammatory activities.
Important categories of MSD by ICF
The musculoskeletal system disorders (MSD) are a group of ailments with high prevalence among the Karen people (Phumthum et al. Citation2020). These diseases accounted for one-third of all use reports of medicinal plants among the Karen (Phumthum et al. Citation2020). There were 23 categories of ailments related to MSD, which were reported from the key informants. The muscle pain category was the most prevalent category of MSD in this study, which agreed with the study of other Karen communities (Kantasrila et al. Citation2020). This ailment relates to the Karen lifestyle, especially their agricultural activities (Kantasrila et al. Citation2020; Phumthum et al. Citation2020), e.g., working in rice fields. Therefore, many species were reported to be used to treat this illness. Moreover, muscle pain was also reported as one of the most prevalent ailments of MSD in other regions around the world such as northern Pakistan (Malik et al. Citation2018), and Spain (Cavero and Calvo Citation2015).
Other ailment categories with high numbers of use reports included flank/axilla symptom/complaint, back symptom/complaint, knee symptom/complaint and joint symptom/complaint NOS. Lumbago was the most common ailment in the flank/axilla symptom/complaint category. Most Karen people have suffered from this ailment (Kantasrila et al. Citation2020; Phumthum et al. Citation2020). The load of hard work in rice fields was the main cause of back pain and lumbago. One of the most prevalent MSD in farmers was pain in the lower back caused by physical activities such as excessive bending, twisting, and carrying of loads (Puntumetakul et al. Citation2011). This result agrees with a study of the prevalence of musculoskeletal pain in Korean farmers, which indicated that low back pain was more frequent than pain in other parts of the body (Min et al. Citation2016). The workload in the rice field also affects other parts of the body such as legs/feet, knees, and shoulders (Rosecrance et al. Citation2006; Min et al. Citation2016).
Phytochemical analysis
Prevalence of phytochemical constituents and pharmacological activities related to MSD
A total of 172 active compounds which are potentially useful for treating MSD are reported. These active compounds possessed the properties which related to anti- inflammatory, analgesic, antinociceptive, anesthetics, anti-arthritic, and antioxidative activities. The most prevalent active compounds included 9-hydroxycalabaxanthone, dihydrovaltrate, and morroniside. 9-Hydroxycalabaxanthone, benzopyrans have been reported for their anti-inflammatory and antioxidative treatments (Gutierrez-Orozco and Failla Citation2013). Dihydrovaltrate is a terpenoid, which is related to anti-inflammatory properties (Subhan et al. Citation2007). Morroniside is an iridoid (monoterpenoid) glycosides, which has been used as an anti-inflammatory agent (Chen et al. Citation2018).
Moreover, there were other prevalent active compounds, including isoacteoside, lithocholic acid, pomiferin, cucurbitacin E, leonuriside A, liriodendrin, and physalin E. Isoacteoside are a hydroxycinnamic acid, which are related to antioxidative and anti-inflammatory activities (Chao-Hsiang et al. Citation2012; Nam et al. Citation2015; Simamora et al. Citation2020). Lithocholic acid is a steroid which has been used as anti-inflammatory activity (Sun et al. Citation2008; Ward et al. Citation2017). Pomiferin is a flavonoid, which is related to antioxidative activity (Bartosikova et al. Citation2007; Mazumder and Rahman Citation2008). Cucurbitacin E is a natural product in member of steroid, which is related to anti-inflammatory and analgesic activities (Peters et al. Citation1997; Abdelwahab et al. Citation2011). Leonuriside A is a phenolic glycoside, which has been used for anti-inflammatory activity (Shrestha et al. Citation2013; Dang et al. Citation2020). Liriodendrin, a lignan glycosides, has been used elsewhere for anti-inflammatory and anti-nociceptive activities (Jung et al. Citation2003). Physalin E is a physalin, which includes anti-inflammatory activity (Pinto et al. Citation2010; Yang et al. Citation2017).
The flavonoids and their glycosides were the main class of active compounds found in this study. They were found in important medicinal plants used for treating MSD. They consist of a large group of polyphenolic compounds having a benzo-γ-pyrone structure and they have been found in many plants (Kumar and Pandey Citation2013). Several studies have documented the pharmacological relevance of flavonoids to their anti-inflammatory, analgesic, and antioxidative activities (Kanadaswami et al. Citation2005; Serafini et al. Citation2010; Barros et al. Citation2011; Carocho and Ferreira Citation2013). Flavonoids can promote human health and lower the risk of diseases (Kumar and Pandey Citation2013). Moreover, the flavonoids are reported to possess anti-inflammatory and analgesic effects by inhibiting the enzyme which is related to inflammation including nitric oxide synthase, cyclooxygenase, and lipoxygenase (Rice-evans et al. Citation1995; Kumar and Pandey Citation2013).
Other active compounds were in the classes of terpenoids, alkaloids, and steroids. Terpenoids are the largest groups of natural compounds, which provide medical benefits for an organism. They are mostly found in plants, especially, in essential oils. Many terpenoids were reported to their anti-inflammatory, antioxidative and analgesic activities (Akihisa et al. Citation2011; Erfanparast et al. Citation2015; Shan et al. Citation2019), which are related to treating the MSD. In this study, many terpenoids were found to be related to treating MSD such as acuminoside (Sharma and Rao Citation2009), bruceine D (Hall et al. Citation1983), agnuside (Pandey et al. Citation2012), grayanotoxin I (Gunduz et al. Citation2014; Sun et al. Citation2019), salannin (Akihisa et al. Citation2011), etc.
Alkaloids are important compounds in many plant species. These compounds have properties related to anti-inflammatory activities (Barbosa-Filho et al. Citation2006). For example, berbamine, berberine, cepharanthine, tetrandine and aconitine have been reported to have anti-inflammatory activity (Wong et al. Citation1992; Barbosa-Filho et al. Citation2006). In this study, many alkaloids were found which in other studies have been demonstrated to be related to anti-inflammatory, analgesic and antioxidative properties, e.g., 3-α(S)-strictosidine (Sakamoto et al. Citation2020), aconine (Li et al. Citation2016), castanospermine (Hong et al. Citation2016), drotaverine (Debski et al. Citation2007; Dash et al. Citation2012), and hirsuteine (Kawakami et al. Citation2011).
Another important phytochemical compound group is steroids. Many steroidal agents are used to treat inflammatory disorders (Vyvey Citation2010; Patel and Savjani Citation2015). Glucocorticoids are steroids, which are mostly used as anti-inflammatory agents (Nunes et al. Citation2020). Extracts from many medicinal plants, which are rich in steroids, are reported elsewhere to be used for their analgesic and anti-inflammatory activities such as Echinops kebericho Mesfin (Asteraceae) (Yimer et al. Citation2020) and Solanum xanthocarpum Schrad (Solanaceae) (Patel and Savjani Citation2015). In this study, many steroidal compounds, which are related to anti-inflammatory activity, were detected, for example, contignasterol (Burgoyne et al. Citation1992; Cheung et al. Citation2016), cortisone (Park et al. Citation2013), cucurbitacin E (Peters et al. Citation1997; Abdelwahab et al. Citation2011), fasciculol C (Ványolós et al. Citation2020), and fluocinonide (Mutasim Citation2006).
Phytochemical compounds and pharmacological activity in important species to treat MSD
The medicinal plants with the highest number of active compounds were Eurycoma longifolia and Flacourtia jangomas. The number of active compounds found in E. longifolia and F. jangomas were one-sixth of total active compounds found in the 20 selected medicinal plants. Eurycoma longifolia was reported for its uses as medicine in many countries around the world (Muhamad et al. Citation2009; Park et al. Citation2014; Baiee et al. Citation2018), especially in Southeast Asia (Muhamad et al. Citation2009). It has been used to treat many illnesses such as malaria, fever (Bhat and Karim Citation2010), gout (Liu et al. Citation2009), and bone pain (Kuo et al. Citation2003). The chemical compounds in this plant were found to possess antioxidative (Lulu et al. Citation2015) and anti-inflammatory activities (Varghese et al. Citation2013). This species had many prevalent compounds, which had pharmacological activities related to MSD such as cis-p-coumaric acid (Pei et al. Citation2016) and vulgarone B (Chung and Shin Citation2009).
Flacourtia jangomas was another well-known species which is used to treat muscle pain among the Karen people (Kamwong Citation2009). This plant has also been used as medicine in India, Cambodia, Laos, Vietnam, and elsewhere in Thailand (Sasi et al. Citation2018). It produces various compounds such as anthocyanin, alkaloids, β-carotene, flavonoids, tannins, saponins, amino acids, and phenolic compounds (Dubey and Pandey Citation2013; Sasi et al. Citation2018). Some of the active chemical constituents that have been reported for many medicinal properties include analgesic, anti-inflammatory, antibacterial, antidiarrheal, antiviral, antioxidant, and anti-amylase activities (Jeyachandran and Mahesh Citation2007).
Other species with high numbers of active compounds were Croton kongensis, Plantago major, Blumea balsamifera, Betula alnoides, Tadehagi triquetrum, Scleropyrum pentandrum, Sambucus javanica and Polygala chinensis. Croton kongensis is a popular medicine among several ethnic groups in Thailand. This species was not only used to treat muscular pain, but also for dysmenorrhea (Salatino et al. Citation2007). The active compounds of this species can inhibit the inflammatory process such as norvaline (Ming et al. Citation2009), vulgarone B (Chung and Shin Citation2009), terretonin (Wu et al. Citation2019), and quercetagetin (Boots et al. Citation2008; Kang et al. Citation2013; Gutiérrez-Venegas et al. Citation2017). In addition, many compounds in this species have been reported to be related with antioxidative and analgesic activities such as benzocaine (Eslamian et al. Citation2016), morroniside (Chen et al. Citation2018), and lithocholic acid (Sun et al. Citation2008).
Plantago major was used to treat many MSD ailments such as rheumatic ailments, bone fractures and muscle pains (Areekun and Onlamun Citation1978; Anderson Citation1993). Extractions from this species had many active compounds related to MSD treatment properties, such as asperuloside (He et al. Citation2018), benzocaine (Eslamian et al. Citation2016), dihydrocapsaicin (Zak et al. Citation2018), piplartine and lithocholic acid (Sun et al. Citation2008; Ward et al. Citation2017).
Blumea balsamifera is a common medicinal plant, which has been used to treat various illnesses among many ethnic groups in Thailand, especially for postpartum recovery of women (Srithi et al. Citation2009; Tangjitman et al. Citation2015). The prevalent compounds found in this species were artemisinin (Kim et al. Citation2015; Shi et al. Citation2015), β-estradiol (Cuzzocrea et al. Citation2000; Song et al. Citation2019), cis-5-caffeoylquinic acid (Ma et al. Citation2011), lithocholic acid (Sun et al. Citation2008; Ward et al. Citation2017), and colforsin (Hayashida et al. Citation2001).
The extracted solution from the bark of Betula alnoides has been used as medicine for a long time in many countries around the world (Rastogi et al. Citation2015). A decoction from the bark of this plant has been used to treat micro-fractures and dislocated bones (Manandhar Citation1995; Rajbhandari Citation2001). The solution of this plant was reported for its anti-inflammatory and antioxidative activities (Sur et al. Citation2002; Ghimire et al. Citation2012). In this study, many active compounds which related to MSD treatment were found, such as benzocaine (Eslamian et al. Citation2016), 5-O-feruloylquinic acid (Erel et al. Citation2011; Sarı et al. Citation2019) and calcitroic acid (Bishop et al. Citation2021).
Tadehagi triquetrum has been used in many Thai folk medicines to treat various ailments including treatments of muscle pain and for improving physical and sexual performances in men (Wisuitiprot et al. Citation2020). It was used in traditional Chinese medicine and reported for its anti-inflammatory activity (Yan et al. Citation2018). The ethanolic and water extracts of this plant were also reported to have potent antioxidant activity (Wisuitiprot et al. Citation2020). In this study, many active compounds related to anti-inflammatory, antioxidative, and analgesic activities were found, e.g., artemisinin (Kim et al. Citation2015; Shi et al. Citation2015), cis-p-coumaric acid (Pei et al. Citation2016), leonuriside A (Shrestha et al. Citation2013; Dang et al. Citation2020), etc.
Some active compounds in Scleropyrum pentandrum were reported to be anti-inflammatory such as ellagic acid (Favarin et al. Citation2013; Kilic et al. Citation2014), castanospermine (Hong et al. Citation2016), cytosine (Amir et al. Citation2007), isofraxidin (Niu et al. Citation2012; Lin et al. Citation2018). Moreover, many active compounds from S. pentandrum were related to antioxidative and analgesic properties such as morroniside (Chen et al. Citation2018) and α-citronellol (Katsukawa et al. Citation2011; Brito et al. Citation2012).
Polygala chinensis was used as a tonic and to treat muscle pain by brewing and drinking it in a water or alcoholic decoction (Areekul et al. Citation2008). Many active compounds in this species were related to anti-inflammation activity such as 9-hydroxycalabaxanthone (Gutierrez-Orozco and Failla Citation2013), leonuriside A (Dang et al. Citation2020), antioxidative activity such as isoacteoside (Simamora et al. Citation2020), fasciculol C (Ványolós et al. Citation2020) and anti-analgesic activity such as astragalin (Qnais et al. Citation2014; Riaz et al. Citation2018).
Another species, with many active compounds was Sambucus javanica, which is used as medicine by several ethnic groups in Thailand (Areekul et al. Citation2008; Inta Citation2008; Kantasrila Citation2016). However, its phytochemical compounds are not well studied yet. In this study, many active compounds were found in S. javanica, which were related to anti-inflammatory, antioxidative and analgesic properties such as 4-guanidinobutanoic acid (Hwang and Jeong Citation2012), 2-hydroxy-1,4-benzoquinone (Petronzi et al. Citation2011), leonuriside A (Shrestha et al. Citation2013; Dang et al. Citation2020), etc.
These promising plants have phytochemical compounds, which related to treat MSD. However, the study about medicinal plants has many concerns such as translational hurdles of phytoconstituents and safety concern. Although medicinal plants play vital roles in disease prevention. There is little scientific evidence to determine the safety and efficacy of most herbal products. There are reports that medicinal plants remedies in the form of complementary and alternative medicine effect adverse reactions among pediatric patients (Lombardi et al. Citation2019). In regard to using medicinal plants, oral administration was the predominant route of administration for most of medicinal plants to treat MSD. However, the oral route of administration needs a high dose of phytomolecule to attain the desired therapeutic plasma concentration (Sharma et al. Citation2004). Many phytochemical compounds have a low percentage of oral bioavailability and have hurdles in clinical translation of molecule due to low solubility and permeability, extensive metabolism, high protein binding, rapid elimination, unstable at physiological pH, poor absorption, etc. (Sayed et al. Citation2019). Therefore, the selection of medicinal plants to study in future clinical tests should consider these hurdles.
Future clinical studies and the road map to the widespread use of medicinal plants to treat MSD for a global patient population
The promising plants such as Sambucus javanica, Betula alnoides, Blumea balsamifera, Plantago major and Flacourtia jangomas and their active components e.g., 9-hydroxycalabaxanthone, dihydrovaltrate and morroniside will be chosen for further clinical investigation. First, we will study in in vitro. The extracts of each plant and pure components will be performed on the critical musculoskeletal system disorders (MSD), e.g., the reduction of prostaglandins (PG) which can cause chronic pain and inflammation. The arachidonic pathway and nuclear factor kappa-B pathway control the production of PG. Therefore, we will focus on related genes and protein expression of this mechanism, especially on cyclooxygenase-2 (COX-2) enzyme. The most effective plant will be further developed into commercial products while the active component will be used as a marker. After that, we will confirm their pharmacological activities, together with their active compounds. The most effective plant will be developed as cream or spray or ointment. Then the effectiveness will be confirmed again in in vitro, in vivo, and clinical studies. The product will be registered for FDA and validated in market in Thailand and in other countries. Use on a global population level might be happening in no more than 6 years.
Conclusions
Medicinal plants used for treating musculoskeletal disorders (MSD) among the Karen were studied in the six villages in Chiang Mai province, Thailand. There were 139 species, 117 genera, 62 families, and 3731 use reports. The phytochemical analysis with (LC-MS/MS) revealed 172 active phytochemical compounds in the 20 examined medicinal species. Most active compounds were flavonoids. The species, which were candidate species in this study, were Sambucus javanica, Betula alnoides, Blumea balsamifera, Plantago major, and Flacourtia jangomas. The occurrence of various active compounds in the 20 most used ethnomedicinal plants validates the traditional knowledge relating to them as remedies for treating MSD. Quantitative method of ethnobotanical study can be used for selecting plants for medicinal development and can be applied for studying other groups of medicinal plants used to treat other disorders in Thailand such as digestive system disorders, infections/infestations disorders, genitourinary system disorders, etc. Moreover, this study provides a valuable information, demonstrating low-cost medicinal plants which are locally available. It is a choice of treatment for the people living in remote areas because medicinal plants can be easily available to poorer segments of the ethnic population. In addition to being a guideline in the field of medicine, it can be used for plant conservation and management.
Acknowledgments
We would like to thank the Karen people in Chiang Mai, Thailand, for sharing their knowledge with us. We are also thankful to the Human Resource Development in Science Project (Science Achievement Scholarship of Thailand, SAST) for supporting the PhD study of Rapeeporn Kantasrila and the Chiang Mai University for partial financial support.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Abdelwahab SI, Hassan LEA, Sirat HM, Yagi SMA, Koko WS, Mohan S, Taha MME, Ahmad S, Chuen CS, Narrima P, et al. 2011. Anti-inflammatory activities of cucurbitacin E isolated from Citrullus lanatus var. citroides: role of reactive nitrogen species and cyclooxygenase enzyme inhibition. Fitoterapia. 82(8): 1190–1197. doi: 10.1016/j.fitote.2011.08.002.
- Afridi R, Khan AU, Khalid S, Shal B, Rasheed H, Ullah MZ, Shehzad O, Kim YS, Khan S. 2019. Anti-hyperalgesic properties of a flavanone derivative poncirin in acute and chronic inflammatory pain models in mice. BMC Pharmacol Toxicol. 20(1): 57. doi: 10.1186/s40360-019-0335-5.
- Afsar T, Khan MR, Razak S, Ullah S, Mirza B. 2015. Antipyretic, anti-inflammatory and analgesic activity of Acacia hydaspica R. Parker and its phytochemical analysis. BMC Complement Altern Med. 15(1): 136. doi: 10.1186/s12906-015-0658-8.
- Agarwal O. 1982. The anti-inflammatory action of nepitrin, a flavonoid. Agents Actions. 12(3): 298–302. doi: 10.1007/BF01965393.
- Agarwal P, Sharma B, Alok S. 2014. Screening of anti-inflammatory and anti-analgesic activity of Convolvulus pluricaulis Choisy. Int J Pharm Sci Res. 5:2458.
- Ahmad W, Ansari HA, Tarannum T. 2010. Curcuma longa. Hippocratic J Unani Med. 5: 179–190.
- Ahn KS, Noh EJ, Cha KH, Kim YS, Lim SS, Shin KH, Jung SH. 2006. Inhibitory effects of irigenin from the rhizomes of Belamcanda chinensis on nitric oxide and prostaglandin E2 production in murine macrophage RAW 264.7 cells. Life Sci. 78(20): 2336–2342. doi: 10.1016/j.lfs.2005.09.041.
- Akihisa T, Takahashi A, Kikuchi T, Takagi M, Watanabe K, Fukatsu M, Fujita Y, Banno N, Tokuda H, Yasukawa K. 2011. The melanogenesis-inhibitory, anti-inflammatory, and chemopreventive effects of limonoids in n-hexane extract of Azadirachta indica A. Juss.(neem) seeds. J Oleo Sci. 60(2): 53–59. doi: 10.5650/jos.60.53.
- Akram M, Syed AS, Kim KA, Lee JS, Chang SY, Kim CY, Bae ON. 2015. Heme oxygenase 1-mediated novel anti-inflammatory activities of Salvia plebeia and its active components. J Ethnopharmacol. 174: 322–330. doi: 10.1016/j.jep.2015.08.028.
- Amir M, Javed S, Kumar H. 2007. Pyrimidine as antiinflammatory agent: a review. Indian J Pharm Sci. 69(3): 337. doi: 10.4103/0250-474X.34540.
- Anderson EF. 1993. Plant and people of the golden triangle: ethnobotany of the hill tribe of the Northern Thailand. Portland, Oregon: Whitman College and Desert Botanical Garden.
- Anh NTT, Nishitani M, Harada S, Yamaguchi M, Kamei K. 2011. A Drosophila model for the screening of bioavailable NADPH oxidase inhibitors and antioxidants. Mol Cell Biochem. 352(1–2): 91–98. doi: 10.1007/s11010-011-0743-3.
- Aniya Y, Fong KF, Naito A, Sakanashi M. 1995. Antioxidative action of the β-adrenoceptor antagonist bopindolol and its metabolite 18-502. Jpn J Pharmacol. 68(3): 323–329. doi: 10.1254/jjp.68.323.
- Apea-Bah FB, Head D, Scales R, Bazylo R, Beta T. 2020. Hydrothermal extraction, a promising method for concentrating phenolic antioxidants from red osier dogwood (Cornus stolonifer) leaves and stems. Heliyon. 6(10): e05158. doi: 10.1016/j.heliyon.2020.e05158.
- Archam N. 2011. Traditional medicinal plants of Tai Yai in Laktaeng village, Wiang Haeng district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University.
- Areekul S, Intorn J, Takheaw S, Nantakaw A. 2008. Wild plant knowledge used in Northern Thailand III (In Thai). Bangkok: Amarin Printing & Publishing Public Company Limited.
- Areekun S, Onlamun A. 1978. Food plants and medicinal plants of ethnic groups in Doi Ang Khang, Chiangmai (In Thai). Kasetsart University: Agricultural development projects.
- Arora C, Tamrakar V. 2017. Gmelina arborea: chemical constituents, pharmacological activities and applications. Int J Phytomed. 9(4): 528–542. doi: 10.5138/09750185.2149.
- Asavachichan S. 2010. Chiang Mai. Bangkok: Saarakadee Press.
- Awaad AS, El-Sayed N, Maitland D, Mabry T. 2006. Phenolic antioxidants from Casimiroa edulis. leaves. Pharm Biol. 44(4): 258–262. doi: 10.1080/13880200600713899.
- Ayumi RR, Mossadeq WMS, Zakaria ZA, Bakhtiar MT, Kamarudin N, Hisamuddin N, Talib M, Sabar AM. 2020. Antinociceptive activity of asiaticoside in mouse models of induced nociception. Planta Med. 86(8): 548–555. doi: 10.1055/a-1144-3663.
- Baiee FH, Wahid H, Rosnina Y, Ariff O, Yimer N, Jeber Z, Salman H, Tarig A, Harighi F. 2018. Impact of Eurycoma longifolia extract on DNA integrity, lipid peroxidation, and functional parameters in chilled and cryopreserved bull sperm. Cryobiology. 80: 43–50. doi: 10.1016/j.cryobiol.2017.12.006.
- Balkrishna A, Thakur P, Varshney A. 2020. Phytochemical profile, pharmacological attributes and medicinal properties of Convolvulus prostrates – a cognitive enhancer herb for the management of neurodegenerative etiologies. Front Pharmacol. 11: 171. doi: 10.3389/fphar.2020.00171.
- Barbosa-Filho JM, Piuvezam MR, Moura MD, Silva MS, Lima KVB, da-Cunha EVL, Fechine IM, Takemura OS. 2006. Anti-inflammatory activity of alkaloids: A twenty-century review. Rev Bras Farmacogn. 16(1): 109–139. doi: 10.1590/S0102-695X2006000100020.
- Barreca D, Laganà G, Leuzzi U, Smeriglio A, Trombetta D, Bellocco E. 2016. Evaluation of the nutraceutical, antioxidant and cytoprotective properties of ripe pistachio (Pistacia vera L., variety Bronte) hulls. Food Chem. 196: 493–502. doi: 10.1016/j.foodchem.2015.09.077.
- Barros L, Cabrita L, Boas MV, Carvalho AM, Ferreira IC. 2011. Chemical, biochemical and electrochemical assays to evaluate phytochemicals and antioxidant activity of wild plants. Food Chem. 127(4):1600–1608. doi: 10.1016/j.foodchem.2011.02.024. [InsertedFromOnline
- Barton K, Randal G, Sagone AL. 1989. The effects of the anti-tumor agent mezerein on the cytotoxic capacity and oxidative metabolism of human blood cells. Invest New Drugs. 7(2–3): 179–188. doi: 10.1007/BF00170855.
- Bartosikova L, Necas J, Bartosik T, Pavlik M, Franova J, Kuzmina G, Luza J. 2007. Examination of the antioxidative and antidiabetic effect of pomiferin in alloxan-induced diabetes mellitus in an experiment (a pilot study). Ceska Slov Farm. 56: 135–140.
- Baydoun E, Bano S, Wahab AT, Jabeen A, Yousuf S, Mesaik A, Smith C, Choudhary MI. 2014. Fungal transformation and T-cell proliferation inhibitory activity of melengestrol acetate and its metabolite. Steroids. 86: 56–61. doi: 10.1016/j.steroids.2014.04.012.
- Beaudoin GA, Facchini PJ. 2014. Benzylisoquinoline alkaloid biosynthesis in opium poppy. Planta. 240(1): 19–32. doi: 10.1007/s00425-014-2056-8.
- Bhat R, Karim A. 2010. Tongkat Ali (Eurycoma longifolia Jack): a review on its ethnobotany and pharmacological importance. Fitoterapia. 81(7): 669–679. doi: 10.1016/j.fitote.2010.04.006.
- Bibi S, Balemba O, Carrion‐Rabanal R, Nyavor Y, Noratto G. 2014. The anti‐inflammatory effects of manniflavanone extracted from Garcinia buchananii bark in obesity‐induced mice model (259.3). Faseb J. 28: 259.
- Bishop LE, Ismailova A, Dimeloe S, Hewison M, White JH. 2021. Vitamin D and immune regulation: antibacterial, antiviral, anti‐inflammatory. JBMR Plus. 5(1): e10405. doi: 10.1002/jbm4.10405.
- Black M, Zacny J, Young C, Klock P, Klafta J, Coalson D, Hill J. 1998. The analgesic effects of remifentanil and alfentanil in healthy volunteers. Anesth Analg. 86(2S):259S. doi: 10.1097/00000539-199802001-00257.
- Boonkorn P. 1997. Ethnobotanical study of Lisu at Sarm-kula village, Wiang Papao, Chiang Rai (In Thai). Chiang Mai, Thailand: Chiang Mai University.
- Boots AW, Wilms LC, Swennen EL, Kleinjans JC, Bast A, Haenen GR. 2008. In vitro and ex vivo anti-inflammatory activity of quercetin in healthy volunteers. Nutrition. 24(7–8): 703–710. doi: 10.1016/j.nut.2008.03.023.
- Borsato ML, Grael CF, Souza GE, Lopes NP. 2000. Analgesic activity of the lignans from Lychnophora ericoides. Phytochemistry. 55(7): 809–813. doi: 10.1016/s0031-9422(00)00388-5.
- Bosi CF, Rosa DW, Grougnet R, Lemonakis N, Halabalaki M, Skaltsounis AL, Biavatti MW. 2013. Pyrrolizidine alkaloids in medicinal tea of Ageratum conyzoides. Rev Bras Farmacogn. 23(3): 425–432. doi: 10.1590/S0102-695X2013005000028.
- Brito RG, Guimarães AG, Quintans JSS, Santos MRV, De Sousa DP, Badaue-Passos D, de Lucca W, Brito FA, Barreto EO, Oliveira AP, et al. 2012. Citronellol, a monoterpene alcohol, reduces nociceptive and inflammatory activities in rodents. J Nat Med. 66(4): 637–644. doi: 10.1007/s11418-012-0632-4.
- Bruchac M. 2014. Indigenous knowledge and traditional knowledge. In: Smith C, editor. Encyclopedia of Global Archaeology. New York: Springer, p. 3814–3824.
- Bulle S, Reddyvari H, Nallanchakravarthula V, Vaddi DR. 2016. Therapeutic potential of Pterocarpus santalinus L.: an update. Pharmacogn Rev. 10(19): 43–49. doi: 10.4103/0973-7847.176575.
- Burgoyne DL, Andersen RJ, Allen TM. 1992. Contignasterol, a highly oxygenated steroid with the unnatural 14. beta. configuration from the marine sponge Petrosia contignata Thiele, 1899. J Org Chem. 57(2): 525–528. doi: 10.1021/jo00028a024.
- Byun MW. 2012. Anti-inflammatory activity of austroinulin from Stevia rebaudiana in LPS-induced RAW264. 7 cells. J Korean Soc Food Sci Nutr. 41(4): 456–461. doi: 10.3746/jkfn.2012.41.4.456.
- Cadena-González AL, Sørensen M, Theilade I. 2013. Use and valuation of native and introduced medicinal plant species in Campo Hermoso and Zetaquira, Boyacá, Colombia. J Ethnobiol Ethnomedicine. 9(1): 1–14.
- Cameron M, Chrubasik S. 2013. Topical herbal therapies for treating osteoarthritis. Cochrane Database Syst Rev. 5: CD010538.
- Carocho M, Ferreira I. 2013. The role of phenolic compounds in the fight against cancer–a review. Anticancer Agents Med Chem. 13(8): 1236–1258. doi: 10.2174/18715206113139990301.
- Caute B, Monsarrat B, Lazorthes Y, Cros J, Bastide R. 1988. The stability of morphine in isobaric and hyperbaric solutions in a drug delivery system. J Pharm Pharmacol. 40(9): 644–645. doi: 10.1111/j.2042-7158.1988.tb05326.x.
- Cavero RY, Calvo MI. 2015. Medicinal plants used for musculoskeletal disorders in Navarra and their pharmacological validation. J Ethnopharmacol. 168: 255–259. doi: 10.1016/j.jep.2015.03.078.
- Chandra SSB, Negara M, Lencho A, Roja, Rani A. 2018. Investigations on biochemical components of Aloe ferox Mill leaf extracts and their antioxidant and anti-inflammatory properties. Int J Dev Res. 8: 18922–18934.
- Changkija S. 1999. Folk medicinal plants of the Nagas in India. Asian Folkl Stud. 58(1): 205–230. doi: 10.2307/1178894.
- Chao-Hsiang C, Yin-Shiou L, Chien MY, Wen-Chi H, Miao-Lin H. 2012. Antioxidant and antihypertensive activities of acteoside and its analogs. Bot Stud. 53: 424–429.
- Chen K, Lu Y, Liu C, Zhang L, Fang Z, Yu G. 2018. Morroniside prevents H2O2 or Aβ1–42-induced apoptosis via attenuating JNK and p38 MAPK phosphorylation. Eur J Pharmacol. 834: 295–304. doi: 10.1016/j.ejphar.2018.07.047.
- Cheung RCF, Ng TB, Wong JH, Chen Y, Chan WY. 2016. Marine natural products with anti-inflammatory activity. Appl Microbiol Biotechnol. 100(4): 1645–1666. doi: 10.1007/s00253-015-7244-3.
- Chin YW, Shin E, Hwang BY, Lee MK. 2011. Antifibrotic constituents from Garcinia mangostana. Nat Prod Commun. 6(9): 1267–1268. 1934578X1100600915.
- Chiu FL, Lin JK. 2005. HPLC analysis of naturally occurring methylated catechins, 33″- and 4″-methyl-epigallocatechin gallate, in various fresh tea leaves and commercial teas and their potent inhibitory effects on inducible nitric oxide synthase in macrophages. J Agric Food Chem. 53(18): 7035–7042. doi: 10.1021/jf0507442.
- Choe KI, Kwon JH, Park KH, Oh MH, Kim MH, Kim HH, Cho SH, Chung EK, Ha SY, Lee MW. 2012. The antioxidant and anti-inflammatory effects of phenolic compounds isolated from the root of Rhodiola sachalinensis A. BOR. Molecules. 17(10): 11484–11494. doi: 10.3390/molecules171011484.
- Choi J, Lee KT, Choi MY, Nam JH, Jung HJ, Park SK, Park HJ. 2005. Antinociceptive anti-inflammatory effect of monotropein isolated from the root of Morinda officinalis. Biol Pharm Bull. 28(10): 1915–1918. doi: 10.1248/bpb.28.1915.
- Choi SB, Park SJ. 2016. Anti-inflammatory effects of 8α-hydroxy pinoresinol isolated from Nardostachys jatamansi on lipopolysaccharide-induced inflammatory response in RAW 264.7 cells. Korean J Herbology. 31(5): 1–6. doi: 10.6116/kjh.2016.31.5.1.
- Chopade AR, Patil PA, Mali SN. 2020. Pharmacological aspects of Phyllanthus fraternus standardized extract (rich in lignans and tannins) as a pain modulator. TOPAINJ. 13(1): 22–34. doi: 10.2174/1876386302013010022.
- Cho JY, Park J, Kim PS, Yoo ES, Baik KU, Park MH. 2001. Savinin, a lignan from Pterocarpus santalinus inhibits tumor necrosis factor-α production and T cell proliferation. Biol Pharm Bull. 24(2): 167–171. doi: 10.1248/bpb.24.167.
- Cho BO, Ryu HW, So Y, Cho JK, Woo HS, Jin CH, Seo KI, Park JC, Jeong IY. 2013. Anti-inflammatory effect of austroinulin and 6-O-acetyl-austroinulin from Stevia rebaudiana in lipopolysaccharide-stimulated RAW264. 7 macrophages. Food Chem Toxicol. 62: 638–644. doi: 10.1016/j.fct.2013.09.011.
- Chung EY, Shin SW. 2009. In vitro anti-inflammatory effects of the essential oil of Artemisia iwayomogi and its main component, vulgarone B. Nat Prod Sci. 15: 229–233.
- Cieza A, Causey K, Kamenov K, Hanson SW, Chatterji S, Vos T. 2020. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 396(10267): 2006–2017. doi: 10.1016/S0140-6736(20)32340-0.
- Collins J, O’Sullivan L. 2010. Psychosocial risk exposures and musculoskeletal disorders across working‐age males and females. Hum Ftrs & Erg Mfg Svc. 20(4): 272–286. doi: 10.1002/hfm.20220.
- Costa MC, Lima TFO, Arcaro CA, Inacio MD, Batista-Duharte A, Carlos IZ, Spolidorio LC, Assis RP, Brunetti IL, Baviera AM. 2020. Trigonelline and curcumin alone, but not in combination, counteract oxidative stress and inflammation and increase glycation product detoxification in the liver and kidney of mice with high-fat diet-induced obesity. J Nutr Biochem. 76: 108303. doi: 10.1016/j.jnutbio.2019.108303.
- Cuzzocrea S, Santagati S, Sautebin L, Mazzon E, Calabrò G, Serraino I, Caputi AP, Maggi A. 2000. 17β-estradiol antiinflammatory activity in carrageenan-induced pleurisy. Endocrinol. 141(4): 1455–1463. doi: 10.1210/endo.141.4.7404.
- Dang HV, Do GH, Ngo PT, Nguyen TD, Le HM. 2020. A new megastigmane glucoside and other constituents from Desmodium gangeticum. J Chem. 2020: 1–4. doi: 10.1155/2020/7416973.
- Dash A, Maiti R, Akantappa Bandakkanavar TK, Arora P. 2012. Intramuscular drotaverine and diclofenac in acute renal colic: a comparative study of analgesic efficacy and safety. Pain Med. 13(3): 466–471. doi: 10.1111/j.1526-4637.2011.01314.x.
- de Melo PF, Kalschne DL, da Silva‐Buzanello RA, Amaral JS, Torquato AS, Corso MP, Falcão HG, Colla E, Ida EI, Canan C. 2020. Cereal bars functionalised with tempeh: nutritional composition, isoflavone content and consumer acceptance. Int J of Food Sci Tech. 55(1): 397–405. doi: 10.1111/ijfs.14384.
- de Menezes Epifanio NM, Cavalcanti LRI, Santos KFD, Duarte PSC, Kachlicki P, Ożarowski M, Riger CJ, de Almeida Chaves DS. 2020. Chemical characterization and in vivo antioxidant activity of parsley (Petroselinum crispum) aqueous extract. Food Funct. 11(6): 5346–5356. doi: 10.1039/d0fo00484g.
- de Padua LS, Bubypraphatsara N, Lemmens RHMJ. 1999. Plant Resources of South-East Asia No. 12(1): medicinal and poisonous plants I. Leiden, The Netherlands: Backhuys Publishers.
- De Souza MM, Bittar M, Cechinel-Filho V, Yunes RA, Messana I, Delle Monache F, Ferrari F. 2000. Antinociceptive properties of morusin, a prenylflavonoid isolated from Morus nigra root bark. Z Naturforsch C J Biosci. 55(3–4): 256–260. doi: 10.1515/znc-2000-3-418.
- Debski R, Niemiec T, Mazurek M, Debska M. 2007. Comparative efficacy and tolerability of drotaverine 80 mg and ibuprofen 400 mg in patients with primary dysmenorrhoea–protocol DOROTA. Ginekol Pol. 78(12): 933–938.
- Dechecchi MC, Nicolis E, Norez C, Bezzerri V, Borgatti M, Mancini I, Rizzotti P, Ribeiro CMP, Gambari R, Becq F, et al. 2008. Anti-inflammatory effect of miglustat in bronchial epithelial cells. J Cyst Fibros. 7(6): 555–565. doi: 10.1016/j.jcf.2008.06.002.
- Deepak M, Handa SS. 2000. Antiinflammatory activity and chemical composition of extracts of Verbena officinalis. Phytother Res. 14(6): 463–465. doi: 10.1002/1099-1573(200009)14:6<463::AID-PTR611>3.0.CO;2-G.
- Department of Social Development and Welfare [DSDW]. 2016. Higland Communities within 20 Provinces of Thailand. Ministry of Social Development and Human Security.
- Dubey N, Pandey V. 2013. Reducing activity of fruits of Flacourtia jangomas (Lour.) Raeusch. Adv Life Sci. 2: 82–83.
- Edenharder R, Grünhage D. 2003. Free radical scavenging abilities of flavonoids as mechanism of protection against mutagenicity induced by tert-butyl hydroperoxide or cumene hydroperoxide in Salmonella typhimurium TA102. Mutat Res. 540(1): 1–18. doi: 10.1016/s1383-5718(03)00114-1.
- Eid O, Ezzat S, Gonaid M, Choucry M. 2018. Crassulaceae (chemistry and pharmacology) – a review. Future J Pharm Sci. 4(2): 234–240. doi: 10.1016/j.fjps.2018.07.003.
- Ercan N, Uludag MO, Agis ER, Demirel-Yilmaz E. 2013. The anti-inflammatory effect of diclofenac is considerably augmented by topical capsaicinoids-containing patch in carrageenan-induced paw oedema of rat. Inflammopharmacology. 21(6): 413–419. doi: 10.1007/s10787-013-0175-7.
- Erel SB, Karaalp C, Bedir E, Kaehlig H, Glasl S, Khan S, Krenn L. 2011. Secondary metabolites of Centaurea calolepis and evaluation of cnicin for anti-inflammatory, antioxidant, and cytotoxic activities. Pharm Biol. 49(8): 840–849. doi: 10.3109/13880209.2010.551538.
- Erfanparast A, Tamaddonfard E, Taati M, Dabbaghi M. 2015. Effects of crocin and safranal, saffron constituents, on the formalin-induced orofacial pain in rats. Avicenna J Phytomed. 5: 392.
- Eslamian L, Borzabadi-Farahani A, Gholami H. 2016. The effect of benzocaine and ketoprofen gels on pain during fixed orthodontic appliance treatment: a randomised, double-blind, crossover trial. Aust Orthod J. 32(1): 64–72. doi: 10.21307/aoj-2020-114.
- Espinosa MM, Bieski ICG, Martins DTO. 2012. Probability sampling design in ethnobotanical surveys of medicinal plants. Rev Bras Farmacogn. 22(6): 1362–1367. doi: 10.1590/S0102-695X2012005000091.
- Farzaei MH, Abbasabadi Z, Ardekani MRS, Rahimi R, Farzaei F. 2013. Parsley: a review of ethnopharmacology, phytochemistry and biological activities. J Tradit Chin Med. 33(6): 815–826. doi: 10.1016/s0254-6272(14)60018-2.
- Favarin DC, Teixeira MM, de Andrade EL, de Freitas Alves C, Chica JEL, Sorgi CA, Faccioli LH, Rogerio AP. 2013. Anti-inflammatory effects of ellagic acid on acute lung injury induced by acid in mice. Mediators Inflamm. 2013: 164202. doi: 10.1155/2013/164202.
- Ferraz CR, Carvalho TT, Manchope MF, Artero NA, Rasquel-Oliveira FS, Fattori V, Casagrande R, Verri WA. 2020. Therapeutic potential of flavonoids in pain and inflammation: mechanisms of action, pre-clinical and clinical data, and pharmaceutical development. Molecules. 25(3): 762. doi: 10.3390/molecules25030762.
- Fiume MM, Bergfeld WF, Belsito DV, Hill RA, Klaassen CD, Liebler DC, Marks JG, Jr, Shank RC, Slaga TJ, Snyder PW, et al. 2018. Safety assessment of Rosmarinus officinalis (Rosemary)-derived ingredients as used in cosmetics. Int J Toxicol. 37(3_suppl): 12S–50S. doi: 10.1177/1091581818800020.
- Fuentes E, Paucar F, Tapia F, Ortiz J, Jimenez P, Romero N. 2018. Effect of the composition of extra virgin olive oils on the differentiation and antioxidant capacities of twelve monovarietals. Food Chem. 243: 285–294. doi: 10.1016/j.foodchem.2017.09.130.
- Fusco D, Colloca G, Monaco MRL, Cesari M. 2007. Effects of antioxidant supplementation on the aging process. Clin Interv Aging. 2: 377.
- Gamdzyk M, Doycheva DM, Kang R, Tang H, Travis ZD, Tang J, Zhang JH. 2020. GW0742 activates miR‐17‐5p and inhibits TXNIP/NLRP3‐mediated inflammation after hypoxic‐ischaemic injury in rats and in PC12 cells. J Cell Mol Med. 24(21): 12318–12330. doi: 10.1111/jcmm.15698.
- Gandigawad P, Poojar B, Hodlur N, Sori RK. 2018. Evaluation of anti-inflammatory activity of ethanolic extract of Gmelina arborea in experimental acute and sub-acute inflammatory models in wistar rats. Int J Basic Clin Pharmacol. 8(1): 128. doi: 10.18203/2319-2003.ijbcp20185170.
- Gao KX, Zhao Q, Wang GR, Yu L, Wu JY, Zhao X. 2020. Isorhynchophylline exerts antinociceptive effects on behavioral hyperalgesia and allodynia in a mouse model of neuropathic pain: evidence of a 5-HT1A receptor-mediated mechanism. Front Pharmacol. 11: 318. doi: 10.3389/fphar.2020.00318.
- Garayev E, Giorgio CD, Herbette G, Mabrouki F, Chiffolleau P, Roux D, Sallanon H, Ollivier E, Elias R, Baghdikian B. 2018. Bioassay-guided isolation and UHPLC-DAD-ESI-MS/MS quantification of potential anti-inflammatory phenolic compounds from flowers of Inula montana L. J Ethnopharmacol. 226: 176–184. doi: 10.1016/j.jep.2018.08.005.
- Ghimire BK, Tamang JP, Yu CY, Jung SJ, Chung IM. 2012. Antioxidant, antimicrobial activity and inhibition of α-glucosidase activity by Betula alnoides Buch. bark extract and their relationship with polyphenolic compounds concentration. Immunopharmacol Immunotoxicol. 34(5): 824–831. doi: 10.3109/08923973.2012.661739.
- Goodla L, Manubolu M, Pathakoti K, Jayakumar T, Sheu JR, Fraker M, Tchounwou PB, Poondamalli PR. 2019. Protective effects of Ammannia baccifera against CCl4-induced oxidative stress in rats. IJERPH. 16(8): 1440. doi: 10.3390/ijerph16081440.
- Guerrero E, Abad A, Montenegro G, Olmo ED, López-Pérez JL, Feliciano AS. 2013. Analgesic and anti-inflammatory activity of podophyllotoxin derivatives. Pharm Biol. 51(5): 566–572. doi: 10.3109/13880209.2012.749921.
- Gülçin İ, Elias R, Gepdiremen A, Taoubi K, Köksal E. 2009. Antioxidant secoiridoids from fringe tree (Chionanthus virginicus L.). Wood Sci Technol. 43(3–4): 195–212. doi: 10.1007/s00226-008-0234-1.
- Gunduz A, Eraydin I, Turkmen S, Kalkan O, Turedi S, Eryigit U, Ayar A. 2014. Analgesic effects of mad honey (grayanotoxin) in mice models of acute pain and painful diabetic neuropathy. Hum Exp Toxicol. 33(2): 130–135. doi: 10.1177/0960327113482693.
- Gupta G, Tarique A. 2013. Prevalence of musculoskeletal disorders in farmers of Kanpur-Rural, India. J Commut Med Health Educ. 3: 249–253.
- Gutierrez-Orozco F, Failla ML. 2013. Biological activities and bioavailability of mangosteen xanthones: a critical review of the current evidence. Nutrients. 5(8): 3163–3183. doi: 10.3390/nu5083163.
- Gutiérrez-Venegas G, Torras-Ceballos A, Gómez-Mora JA, Fernández-Rojas B. 2017. Luteolin, quercetin, genistein and quercetagetin inhibit the effects of lipopolysaccharide obtained from Porphyromonas gingivalis in H9c2 cardiomyoblasts. Cell Mol Biol Lett. 22(1): 19. doi: 10.1186/s11658-017-0047-z.
- Hadjipavlou-Litina D, Garnelis T, Athanassopoulos CM, Papaioannou D. 2009. Kukoamine A analogs with lipoxygenase inhibitory activity. J Enzyme Inhib Med Chem. 24(5): 1188–1193. doi: 10.1080/14756360902779193.
- Hall I, Lee K, Imakura Y, Okano M, Johnson A. 1983. Anti-inflammatory agents III: structure–activity relationships of brusatol and related quassinoids. J Pharm Sci. 72(11): 1282–1284. doi: 10.1002/jps.2600721111.
- Hamdi OAA, Ye LJ, Kamarudin MNA, Hazni H, Paydar M, Looi CY, Shilpi JA, Kadir HA, Awang K. 2015. Neuroprotective and antioxidant constituents from Curcuma zedoaria rhizomes. Rec Nat Prod. 9: 349–355.
- Han X, Pan J, Ren D, Cheng Y, Fan P, Lou H. 2008. Naringenin-7-O-glucoside protects against doxorubicin-induced toxicity in H9c2 cardiomyocytes by induction of endogenous antioxidant enzymes. Food Chem Toxicol. 46(9): 3140–3146. doi: 10.1016/j.fct.2008.06.086.
- Hartvigsen J, Hancock MJ, Kongsted A, Louw Q, Ferreira ML, Genevay S, Hoy D, Karppinen J, Pransky G, Sieper J, et al. 2018. What low back pain is and why we need to pay attention. Lancet. 391(10137): 2356–2367. doi: 10.1016/S0140-6736(18)30480-X.
- Hayashida N, Chihara S, Tayama E, Takaseya T, Enomoto N, Kawara T, Aoyagi S. 2001. Antiinflammatory effects of colforsin daropate hydrochloride, a novel water-soluble forskolin derivative. Ann Thorac Surg. 71(6): 1931–1938. doi: 10.1016/s0003-4975(01)02531-0.
- He J, Lu X, Wei T, Dong Y, Cai Z, Tang L, Liu M. 2018. Asperuloside and asperulosidic acid exert an anti-inflammatory effect via suppression of the NF-κB and MAPK signaling pathways in LPS-induced RAW 264.7 macrophages. Int J Mol Sci. 19(7): 2027. doi: 10.3390/ijms19072027.
- Heinrich M, Ankli A, Frei B, Weimann C, Sticher O. 1998. Medicinal plants in Mexico: healers’ consensus and cultural importance. Soc Sci Med. 47(11): 1859–1871. doi: 10.1016/s0277-9536(98)00181-6.
- Herraiz T, Galisteo J. 2002. Tetrahydro-β-carboline alkaloids that occur in foods and biological systems act as radical scavengers and antioxidants in the ABTS assay. Free Radic Res. 36(8): 923–928. doi: 10.1080/1071576021000005762.
- Hignett S, Fray M. 2010. Manual handling in healthcare. Proceedings of the 1st Conference of the Federation of the European Ergonomics Societies [FEES], October 10–12; Bruges, Belgium.
- Hong YP, Chen C, Guo WY, Zhao L, Mei FC, Xiang MW, Wang WX. 2016. Effects of castanospermine on inflammatory response in a rat model of experimental severe acute pancreatitis. Arch Med Res. 47(6): 436–445. doi: 10.1016/j.arcmed.2016.11.007.
- Hossain CF, Al-Amin M, Sayem ASM, Siragee IH, Tunan AM, Hassan F, Kabir MM, Sultana GNN. 2015. Antinociceptive principle from Curcuma aeruginosa. BMC Complement Altern Med. 15(1): 191. doi: 10.1186/s12906-015-0720-6.
- Huang SS, Chiu CS, Chen HJ, Hou WC, Sheu MJ, Lin YC, Shie PH, Huang GJ. 2011. Antinociceptive activities and the mechanisms of anti-inflammation of asiatic acid in mice. Evid Based Complement Alternat Med. 2011: 895857–895810. doi: 10.1155/2011/895857.
- Hussan F, Mansor AS, Hassan SN, Kamaruddin TNE, Tasnim TN, Budin SB, Othman F. 2015. Anti-inflammatory property of Plantago major leaf extract reduces the inflammatory reaction in experimental acetaminophen-induced liver injury. Evid Based Complement Alternat Med. 2015: 347861–347867. doi: 10.1155/2015/347861.
- Hutasingha P. 2015. Ethnobotany of Akha in Mae La-ngong village, Namphrae subdistrict, Phrao district, Chiang Mai province (In Thai). Chiang Mai, Thailand: Chiang Mai University.
- Hwang IY, Jeong CS. 2012. Inhibitory effects of 4-guanidinobutyric acid against gastric lesions. Biomol Ther (Seoul). 20(2): 239–244. doi: 10.4062/biomolther.2012.20.2.239.
- Hwang SJ, Lee HJ. 2015. Phenyl-β-d-glucopyranoside exhibits anti-inflammatory activity in lipopolysaccharide-activated RAW 264.7 cells. Inflammation. 38(3): 1071–1079. doi: 10.1007/s10753-014-0072-2.
- Ibrahim NA, El-Gengaihi S, Motawe H, Riad S. 2007. Phytochemical and biological investigation of Stevia rebaudiana Bertoni; 1-labdane-type diterpene. Eur Food Res Technol. 224(4): 483–488. doi: 10.1007/s00217-006-0400-3.
- Iizuka A, Iijima OT, Kondo K, Itakura H, Yoshie F, Miyamoto H, Kubo M, Higuchi M, Takeda H, Matsumiya T. 2004. Evaluation of Rhubarb using antioxidative activity as an index of pharmacological usefulness. J Ethnopharmacol. 91(1): 89–94. doi: 10.1016/j.jep.2003.11.021.
- Inta A. 2008. Ethnobotany and crop diversity of Tai Lue and Akha communities in the upper northern Thailand and the Xishuangbanna Dai autonomous prefecture, China. Chiang Mai, Thailand: Chiang Mai University.
- Inta A. 2014. [A Comparative study of ethnobotany of ethnic groups in Pang Mapha district, Maehongson provice]. Chiang Mai, Thailand: Department of Biology, Faculty of Science, Chiang Mai University. Thai.
- Inta A, Pongamornkul W. 2015a. [Study and survey of medicinal plants and medicinal plants utilization of the community: sub-project 3]. Chiang Mai, Thailand: Highland Research and Development Institute (Public Organization). Thai.
- Inta A, Pongamornkul W. 2015b. [Study of plant diversity and indigenous botanicals of the community: sub-project 2]. Chiang Mai, Thailand: Highland Research and Development Institute (Public Organization). Thai.
- Inta A, Trisonthi C, Pongamornkul W. 2011a. Ethnobotany of Tai Yai in Chiang Dao district and Wiang Haeng district, Chiang Mai province. Chiang Mai, Thailand: Biology department, Science Faculty, Chiang Mai University. Thai.
- Inta A, Trisonthi C, Trisonthi P. 2011b. An analysis of knowledge on the utilization of local plants of Chiang Mai University Hariphunchai Education Centre, Lamphun Province, Chiang Mai University. Chiang Mai, Thailand: Department of Biology, Faculty of Science, Chiang Mai University.
- Islam S, Nagasaka R, Ohara K, Hosoya T, Ozaki H, Ushio H, Hori M. 2011. Biological abilities of rice bran-derived antioxidant phytochemicals for medical therapy. Curr Top Med Chem. 11(14): 1847–1853. doi: 10.2174/156802611796235099.
- Islam MS, Yoshida H, Matsuki N, Ono K, Nagasaka R, Ushio H, Guo Y, Hiramatsu T, Hosoya T, Murata T, et al. 2009. Antioxidant, free radical–scavenging, and NF-κB–inhibitory activities of phytosteryl ferulates: structure–activity studies. J Pharmacol Sci. 111(4): 328–337. doi: 10.1254/jphs.09146fp.
- Jeyachandran R, Mahesh A. 2007. Enumeration of antidiabetic herbal flora of Tamil Nadu. Research J of Medicinal Plant. 1(4): 144–148. doi: 10.3923/rjmp.2007.144.148.
- Jin M, Sun CY, Zang BX. 2016. Hydroxysafflor yellow A attenuate lipopolysaccharide-induced endothelium inflammatory injury. Chin J Integr Med. 22(1): 36–41. doi: 10.1007/s11655-015-1976-x.
- Ji Y, Yin W, Liang Y, Sun L, Yin Y, Zhang W. 2020. Anti-inflammatory and anti-oxidative activity of indole-3-acetic acid involves induction of HO-1 and neutralization of free radicals in RAW264.7 Cells. Int J Mol Sci. 21(5): 1579. doi: 10.3390/ijms21051579.
- Ji L, Yuan Y, Luo L, Chen Z, Ma X, Ma Z, Cheng L. 2012. Physalins with anti-inflammatory activity are present in Physalis alkekengi var. franchetii and can function as Michael reaction acceptors. Steroids. 77(5): 441–447. doi: 10.1016/j.steroids.2011.11.016.
- Jolayemi A, Ojewole J. 2013. Comparative anti-inflammatory properties of capsaicin and ethylaacetate extract of Capsicum frutescens Linn [Solanaceae] in rats. Afr Health Sci. 13(2): 357–361. doi: 10.4314/ahs.v13i2.23.
- Jung HJ, Park HJ, Kim RG, Shin KM, Ha J, Choi JW, Kim HJ, Lee YS, Lee KT. 2003. In vivo anti-inflammatory and antinociceptive effects of liriodendrin isolated from the stem bark of Acanthopanax senticosus. Planta Med. 69(7): 610–616. doi: 10.1055/s-2003-41127.
- Junkhonkaen J. 2012. Ethnobotany of Ban Bowee, Amphoe Suan Phueng. Changwat Ratchaburi. Bangkok, Thailand: Kasetsart University. Thai.
- Junsongduang A. 2013. Roles and importance of sacred forest in biodiversity conservation in Mae Chaem District, Chiang Mai Province. Chiang Mai, Thailand: Chiang Mai University.
- Junsongduang A, Kasemwan W, Lumjoomjung S, Sabprachai W, Tanming W, Balslev H. 2020. Ethnomedicinal knowledge of traditional healers in Roi Et, Thailand. Plants. 9(9): 1177. doi: 10.3390/plants9091177.
- Kaewsangsai S. 2017. [Ethnobotany of Karen in the royal project extended area Khun Tuen Noi village, Omkoi district, Chiang Mai province. Chiang Mai, Thailand. Chiang Mai University. Thai.
- Kamwong K. 2009. Ethnobotany of Karens at Ban Mai Sawan and Ban Huay Pu Ling, Ban Luang sub-district, Chom Thong district. Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Kanadaswami C, Lee LT, Lee PPH, Hwang JJ, Ke FC, Huang YT, Lee MT. 2005. The antitumor activities of flavonoids. In Vivo. 19: 895–909.
- Kang GJ, Han SC, Kang NJ, Koo DH, Park DB, Eun SY, Kang HK, Yoo ES. 2014. Quercetagetin inhibits macrophage‐derived chemokine in H a C a T human keratinocytes via the regulation of signal transducer and activator of transcription 1, suppressor of cytokine signalling 1 and transforming growth factor‐β1. Br J Dermatol. 171(3): 512–523. doi: 10.1111/bjd.12938.
- Kang GJ, Han SC, Ock JW, Kang HK, Yoo ES. 2013. Anti-inflammatory effect of quercetagetin, an active component of immature Citrus unshiu, in HaCaT human keratinocytes. Biomol Ther (Seoul). 21(2): 138–145. doi: 10.4062/biomolther.2013.001.
- Kang H, Ku SK, Jung B, Bae JS. 2015. Anti-inflammatory effects of vicenin-2 and scolymoside in vitro and in vivo. Inflamm Res. 64(12): 1005–1021. doi: 10.1007/s00011-015-0886-x.
- Kang MY, Lee MJ, Chung H, Shin DH, Youn KW, Im SH, Chae HS, Lee KS. 2016. Musculoskeletal disorders and agricultural risk factors among Korean farmers. J Agromedicine. 21(4): 353–363. doi: 10.1080/1059924X.2016.1178612.
- Kantasrila R. 2016. [Ehtnobotany of Karen at Ban Wa Do Kro, Mae Song sub-district, Tha Song Yang district, Tak province]. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Kantasrila R, Pandith H, Balslev H, Wangpakapattanawong P, Panyadee P, Inta A. 2020. Medicinal plants for treating musculoskeletal disorders among Karen in Thailand. Plants. 9(7): 811. doi: 10.3390/plants9070811.
- Kanwal N, Rasul A, Hussain G, Anwar H, Shah MA, Sarfraz I, Riaz A, Batool R, Shahbaz M, Hussain A, et al. 2020. Oleandrin: a bioactive phytochemical and potential cancer killer via multiple cellular signaling pathways. Food Chem Toxicol. 143: 111570. doi: 10.1016/j.fct.2020.111570.
- Kaswala R, Patel V, Chakraborty M, Kamath J. 2012. Phytochemical and pharmacological profile of Gmelina arborea: an overview. Int Res J Pharm. 3: 61–64.
- Katsukawa M, Nakata R, Koeji S, Hori K, Takahashi S, Inoue H. 2011. Citronellol and geraniol, components of rose oil, activate peroxisome proliferator-activated receptor α and γ and suppress cyclooxygenase-2 expression. Biosci Biotechnol Biochem. 75(5): 1010–1012. doi: 10.1271/bbb.110039.
- Kaur AK, Kumar S, Sharma PK. 2017. Hepatoprotective activity of Inula cappa DC. Aqueous extract against carbon tetrachloride induced hepatotoxicity in Wistar rats. Int Res J Pharm. 8(1): 14–19. doi: 10.7897/2230-8407.08013.
- Kawakami Z, Kanno H, Ikarashi Y, Kase Y. 2011. Yokukansan, a kampo medicine, protects against glutamate cytotoxicity due to oxidative stress in PC12 cells. J Ethnopharmacol. 134(1): 74–81. doi: 10.1016/j.jep.2010.11.063.
- Khaled M, Jiang ZZ, Zhang LY. 2013. Deoxypodophyllotoxin: a promising therapeutic agent from herbal medicine. J Ethnopharmacol. 149(1): 24–34. doi: 10.1016/j.jep.2013.06.021.
- Khuankaew S, Srithi K, Tiansawat P, Jampeetong A, Inta A, Wangpakapattanawong P. 2014. Ethnobotanical study of medicinal plants used by Tai Yai in Northern Thailand. J Ethnopharmacol. 151(2): 829–838. doi: 10.1016/j.jep.2013.11.033.
- Kilic I, Yeşiloğlu Y, Bayrak Y. 2014. Spectroscopic studies on the antioxidant activity of ellagic acid. Spectrochim Acta A Mol Biomol Spectrosc. 130: 447–452. doi: 10.1016/j.saa.2014.04.052.
- Kim WS, Choi WJ, Lee S, Kim WJ, Lee DC, Sohn UD, Shin HS, Kim W. 2015. Anti-inflammatory, antioxidant and antimicrobial effects of artemisinin extracts from Artemisia annua L. Korean J Physiol Pharmacol. 19(1): 21–27. doi: 10.4196/kjpp.2015.19.1.21.
- Kim JB, Han AR, Park EY, Kim JY, Cho W, Lee J, Seo EK, Lee KT. 2007. Inhibition of LPS-induced iNOS, COX-2 and cytokines expression by poncirin through the NF-κB inactivation in RAW 264.7 macrophage cells. Biol Pharm Bull. 30(12): 2345–2351. doi: 10.1248/bpb.30.2345.
- Kim GN, Jang HD. 2011. Flavonol content in the water extract of the mulberry (Morus alba L.) leaf and their antioxidant capacities. J Food Sci. 76(6): C869–C873. doi: 10.1111/j.1750-3841.2011.02262.x.
- Kim SK, Kim HJ, Choi SE, Park KH, Choi HK, Lee MW. 2008. Anti-oxidative and inhibitory activities on nitric oxide (NO) and prostaglandin E 2 (COX-2) production of flavonoids from seeds of Prunus tomentosa Thunberg. Arch Pharm Res. 31(4): 424–428. doi: 10.1007/s12272-001-1174-9.
- Kim NM, Kim J, Chung HY, Choi JS. 2000. Isolation of luteolin 7-O-rutinoside and esculetin with potential antioxidant activity from the aerial parts of Artemisia montana. Arch Pharm Res. 23(3): 237–239. doi: 10.1007/BF02976451.
- Kim KW, Kim HJ, Sohn JH, Yim JH, Kim YC, Oh H. 2018. Anti-neuroinflammatory effect of 6, 8, 1′-tri-O-methylaverantin, a metabolite from a marine-derived fungal strain Aspergillus sp., via upregulation of heme oxygenase-1 in lipopolysaccharide-activated microglia. Neurochem Int. 113: 8–22. doi: 10.1016/j.neuint.2017.11.010.
- Kimura M, Kobayashi S, Luo B, Kimura I. 1991. Selective inhibition by magnosalin and magnoshinin, compounds from” Shin-i” (Flos magnoliae), of adjuvant-induced angiogenesis and granuloma formation in the mouse pouch. Agents Actions Suppl. 32: 197–201. doi: 10.1007/978-3-0348-7405-2_27.
- Kim MJ, Wang HS, Lee MW. 2020. Anti-inflammatory effects of fermented bark of Acanthopanax sessiliflorus and its isolated compounds on lipopolysaccharide-treated RAW 264.7 macrophage cells. Evid Based Complement Alternat Med. 2020: 6749425–6749410. doi: 10.1155/2020/6749425.
- Kiplimo JJ, Islam MS, Koorbanally NA. 2011. A novel flavonoid and furoquinoline alkaloids from Vepris glomerata and their antioxidant activity. Nat Prod Commun. 6(12): 1847–1850. 1934578X1100601215.
- Koley H, Howlader DR, Bhaumik U. 2019. Assessment of antimicrobial activity of different phytochemicals against enteric diseases in different animal models. In: Khan MSA, Ahmad I, Chattopadhyay D, editors. New look to phytomedicine. London: Academic Press, p. 563–580.
- Kotakadi VS. 2014. Anti-inflammatory effect of Basella rubra on oxazolone-induced colitis in rat. AJPCT. 2: 832–841.
- Kremen C, Raymond I, Lance K. 1998. An interdisciplinary tool for monitoring conservation impacts in Madagascar. Conserv Biol. 12(3): 549–563. doi: 10.1111/j.1523-1739.1998.96374.x.
- Kudou S, Fleury Y, Welti D, Magnolato D, Uchida T, Kitamura K, Okubo K. 1991. Malonyl isoflavone glycosides in soybean seeds (Glycine max Merrill). Agric Biol Chem. 55(9): 2227–2233. doi: 10.1271/bbb1961.55.2227.
- Kumar KS, Hsieh HW, Wang SY. 2010a. Anti-inflammatory effect of lucidone in mice via inhibition of NF-κB/MAP kinase pathway. Int Immunopharmacol. 10(4): 385–392. doi: 10.1016/j.intimp.2009.12.013.
- Kumar R, Krishan P, Swami G, Kaur P, Shah G, Kaur A. 2010b. Pharmacognostical investigation of Cymbopogon citratus (DC) Stapf. Der Pharm Lett. 2: 181–189.
- Kumar S, Pandey AK. 2013. Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal. 2013: 162750–162716. doi: 10.1155/2013/162750.
- Kuo P-C, Shi L-S, Damu AG, Su C-R, Huang C-H, Ke C-H, Wu J-B, Lin A-J, Bastow KF, Lee K-H, et al. 2003. Cytotoxic and antimalarial β-carboline alkaloids from the roots of Eurycoma longifolia. J Nat Prod. 66(10): 1324–1327. doi: 10.1021/np030277n.
- Küpeli E, Erdemoğlu N, Yeşilada E, Sener B. 2003. Anti-inflammatory and antinociceptive activity of taxoids and lignans from the heartwood of Taxus baccata L. J Ethnopharmacol. 89(2–3): 265–270. doi: 10.1016/j.jep.2003.09.005.
- Küpeli E, Harput US, Varel M, Yesilada E, Saracoglu I. 2005. Bioassay-guided isolation of iridoid glucosides with antinociceptive and anti-inflammatory activities from Veronica anagallis-aquatica L. J Ethnopharmacol. 102(2): 170–176. doi: 10.1016/j.jep.2005.05.042.
- Lai CS, Lee JH, Ho CT, Liu CB, Wang JM, Wang YJ, Pan MH. 2009. Rosmanol potently inhibits lipopolysaccharide-induced iNOS and COX-2 expression through downregulating MAPK, NF-κB, STAT3 and C/EBP signaling pathways. J Agric Food Chem. 57(22): 10990–10998. doi: 10.1021/jf9025713.
- Landberg R, Sunnerheim K, Dimberg LH. 2020. Avenanthramides as lipoxygenase inhibitors. Heliyon. 6(6): e04304. doi: 10.1016/j.heliyon.2020.e04304.
- Lanhers MC, Fleurentin J, Mortier F, Vinche A, Younos C. 1992. Anti-inflammatory and analgesic effects of an aqueous extract of Harpagophytum procumbens. Planta Med. 58(2): 117–123. doi: 10.1055/s-2006-961411.
- Laville I, Mercier L, Chachaty E, Bourget P, Paci A. 2005. Shelf-lives of morphine and pethidine solutions stored in patient-controlled analgesia devices: physico-chemical and microbiological stability study. Pathol Biol (Paris). 53(4): 210–216. doi: 10.1016/j.patbio.2004.09.007.
- Lawal A. 2016. Phytochemical, Proximate and mineral composition of Gmelina arborea fruits (White Teek). Fountain J Nat Appl Sci. 5: 12–15.
- Lee IC, Bae JS. 2016. Anti-inflammatory effects of vicenin-2 and scolymoside on polyphosphate-mediated vascular inflammatory responses. Inflamm Res. 65(3):203–212. doi: 10.1007/s00011-015-0906-x.
- Lee S, Do SG, Kim SY, Kim J, Jin Y, Lee CH. 2012. Mass spectrometry-based metabolite profiling and antioxidant activity of Aloe vera (Aloe barbadensis Miller) in different growth stages. J Agric Food Chem. 60(45): 11222–11228. doi: 10.1021/jf3026309.
- Lee HJ, Kim KW. 2012. Anti-inflammatory effects of arbutin in lipopolysaccharide-stimulated BV2 microglial cells. Inflamm Res. 61(8): 817–825. doi: 10.1007/s00011-012-0474-2.
- Lee IA, Lee JH, Baek NI, Kim DH. 2005. Antihyperlipidemic effect of crocin isolated from the fructus of Gardenia jasminoides and its metabolite crocetin. Biol Pharm Bull. 28(11): 2106–2110. doi: 10.1248/bpb.28.2106.
- Lee SJ, Shin EJ, Son KH, Chang HW, Kang SS, Kim HP. 1995. Anti-inflammatory activity of the major constituents of Lonicera japonica. Arch Pharm Res. 18(2):133–135. doi: 10.1007/BF02979147.
- Lee SH, Son MJ, Ju HK, Lin CX, Moon TC, Choi HG, Son JK, Chang HW. 2004. Dual inhibition of cyclooxygenases-2 and 5-lipoxygenase by deoxypodophyllotoxin in mouse bone marrow-derived mast cells. Biol Pharm Bull. 27(6): 786–788. doi: 10.1248/bpb.27.786.
- Lee M, Zhao H, Liu X, Liu D, Chen J, Li Z, Chu S, Kou X, Liao S, Deng Y. 2020. Protective effect of hydroxysafflor yellow A on nephropathy by attenuating oxidative stress and inhibiting apoptosis in induced type 2 Diabetes in rat. Oxid Med Cell Longev. 2020: 7805393.
- Li N, Cao L, Cheng Y, Meng ZQ, Tang ZH, Liu WJ, Wang ZZ, Ding G, Xiao W. 2014. In vivo anti-inflammatory and analgesic activities of strictosamide from Nauclea officinalis. Pharm Biol. 52(11): 1445–1450. doi: 10.3109/13880209.2014.895910.
- Li D, Chen J, Ye J, Zhai X, Song J, Jiang C, Wang J, Zhang H, Jia X, Zhu F. 2017a. Anti-inflammatory effect of the six compounds isolated from Nauclea officinalis Pierrc ex Pitard, and molecular mechanism of strictosamide via suppressing the NF-κB and MAPK signaling pathway in LPS-induced RAW 264.7 macrophages. J Ethnopharmacol. 196: 66–74. doi: 10.1016/j.jep.2016.12.007.
- Li Y-J, Guo Y, Yang Q, Weng X-G, Yang L, Wang Y-J, Chen Y, Zhang D, Li Q, Liu X-C, et al. 2015. Flavonoids casticin and chrysosplenol D from Artemisia annua L. inhibit inflammation in vitro and in vivo. Toxicol Appl Pharmacol. 286(3): 151–158. doi: 10.1016/j.taap.2015.04.005.
- Li X, Jiang C, Zhu W. 2017b. Crocin reduces the inflammation response in rheumatoid arthritis. Biosci Biotechnol Biochem. 81(5): 891–898. doi: 10.1080/09168451.2016.1263145.
- Li XJ, Kim KW, Oh H, Liu XQ, Kim YC. 2019c. Chemical constituents and an antineuroinflammatory lignan, savinin from the roots of Acanthopanax henryi. Evid Based Complement Alternat Med. 2019:1856294.
- Li X, Lin J, Chen B, Xie H, Chen D. 2018. Antioxidant and cytoprotective effects of kukoamines A and B: comparison and positional isomeric effect. Molecules. 23(4): 973. doi: 10.3390/molecules23040973.
- Li C, Liu K, Liu S, Aerqin Q, Wu XJ. 2020a. Role of ginkgolides in inflammatory immune response of neurological diseases: a review of current literatures. Front Syst Neurosci. 14: 45. doi: 10.3389/fnsys.2020.00045.
- Li P, Lv B, Jiang X, Wang T, Ma X, Chang N, Wang X, Gao X. 2016. Identification of NF-κB inhibitors following Shenfu injection and bioactivity-integrated UPLC/Q-TOF-MS and screening for related anti-inflammatory targets in vitro and in silico. J Ethnopharmacol. 194: 658–667. doi: 10.1016/j.jep.2016.10.052.
- Lim C, Park JG, Kang KB, Suh YG. 2019. Identification and semi-synthesis of 3-O-protocatechuoylceanothic acid, a novel and natural GPR120 agonist. Molecules. 24(19): 3487. doi: 10.3390/molecules24193487.
- Lin J, Li X, Qi W, Yan Y, Chen K, Xue X, Xu X, Feng Z, Pan X. 2018. Isofraxidin inhibits interleukin-1β induced inflammatory response in human osteoarthritis chondrocytes. Int Immunopharmacol. 64: 238–245. doi: 10.1016/j.intimp.2018.09.003.
- Li Y, Shi J, Sun X, Li Y, Duan Y, Yao H. 2020c. Theaflavic acid from black tea protects PC12 cells against ROS-mediated mitochondrial apoptosis induced by OGD/R via activating Nrf2/ARE signaling pathway. J Nat Med. 74(1): 238–246. doi: 10.1007/s11418-019-01333-4.
- Li C, Tan F, Yang J, Yang Y, Gou Y, Li S, Zhao X. 2019a. Antioxidant effects of Apocynum venetum tea extracts on d-galactose-induced aging model in mice. Antioxidants. 8(9): 381. doi: 10.3390/antiox8090381.
- Liu Y, Dao Z, Yang C, Liu Y, Long C. 2009. Medicinal plants used by Tibetans in Shangri-la, Yunnan, China. J Ethnobiol Ethnomed. 5: 15.
- Liu B, Ma Y, Liu Y, Yang Z, Zhang L. 2014. Ultrasonic-assisted extraction and antioxidant activity of flavonoids from Adinandra nitida leaves. Trop J Pharm Res. 12(6):1045–1051. doi: 10.4314/tjpr.v12i6.27.
- Liu Y, Tong J. 2005. Analgesic effect of nudicauline and its mechanism. Chinese J Tissue Eng Res. 9: 238–239.
- Li G, Yang L, Feng L, Yang J, Li Y, AJ, Li D, Xu Y, Gao Y, Li J. 2020b. Syringaresinol protects against type 1 diabetic cardiomyopathy by alleviating inflammation responses, cardiac fibrosis, and oxidative stress. Mol Nutr Food Res. 64:2000231.
- Li MY, Zhang ZH, Wang Z, Zuo HX, Wang JY, Xing Y, Jin CH, Xu GH, Piao LX, Ma J, et al. 2019b. Convallatoxin protects against dextran sulfate sodium-induced experimental colitis in mice by inhibiting NF-κB signaling through activation of PPARγ. Pharmacol Res. 147: 104355. doi: 10.1016/j.phrs.2019.104355.
- Lombardi N, Crescioli G, Bettiol A, Menniti-Ippolito F, Maggini V, Gallo E, Mugelli A, Vannacci A, Firenzuoli F. 2019. Safety of complementary and alternative medicine in children: a 16-years retrospective analysis of the Italian Phytovigilance system database. Phytomedicine. 61: 152856. doi: 10.1016/j.phymed.2019.152856.
- Lotito SB, Zhang WJ, Yang CS, Crozier A, Frei B. 2011. Metabolic conversion of dietary flavonoids alters their anti-inflammatory and antioxidant properties. Free Radic Biol Med. 51(2): 454–463. doi: 10.1016/j.freeradbiomed.2011.04.032.
- Luangwilai T, Norkaew S, Siriwong W. 2014. Factors associated with musculoskeletal disorders among rice farmers: cross sectional study in Tarnlalord sub-district, Phimai district, Nakhonratchasima Province, Thailand. J Health Res. 28: s 85–S91.
- Luetragoon T, Pankla Sranujit R, Noysang C, Thongsri Y, Potup P, Suphrom N, Nuengchamnong N, Usuwanthim K. 2020. Bioactive compounds in Moringa oleifera Lam. leaves inhibit the pro-inflammatory mediators in lipopolysaccharide-induced human monocyte-derived macrophages. Molecules. 25(1): 191. doi: 10.3390/molecules25010191.
- Lulu T, Park SY, Ibrahim R, Paek KY. 2015. Production of biomass and bioactive compounds from adventitious roots by optimization of culturing conditions of Eurycoma longifolia in balloon-type bubble bioreactor system. J Biosci Bioeng. 119(6): 712–717. doi: 10.1016/j.jbiosc.2014.11.010.
- Luo XJ, Peng J, Li YJ. 2011. Recent advances in the study on capsaicinoids and capsinoids. Eur J Pharmacol. 650(1): 1–7. doi: 10.1016/j.ejphar.2010.09.074.
- Luo L, Zhou J, Zhao H, Fan M, Gao W. 2019. The anti-inflammatory effects of formononetin and ononin on lipopolysaccharide-induced zebrafish models based on lipidomics and targeted transcriptomics. Metabolomics. 15(12):153. doi: 10.1007/s11306-019-1614-2.
- Ma C, Dastmalchi K, Whitaker BD, Kennelly EJ. 2011. Two new antioxidant malonated caffeoylquinic acid isomers in fruits of wild eggplant relatives. J Agric Food Chem. 59(17): 9645–9651. doi: 10.1021/jf202028y.
- Mahawongsanan A. 2008. [Change of herbal plants utilization of the Pga K’nyau: a case study of Ban Huay Som Poy, Mae Tia Watershed, Chom Thong district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Mahmood A, Mahmood A, Malik RN. 2012. Indigenous knowledge of medicinal plants from Leepa valley. J Ethnopharmacol. 143(1): 338–346.
- Mahomoodally M, Sadeer N, Suroowan S, Jugreet S, Lobine D, Rengasamy K. 2021. Ethnomedicinal, phytochemistry, toxicity and pharmacological benefits of poison bulb–Crinum asiaticum L. S Afr J Bot. 136: 16–29. doi: 10.1016/j.sajb.2020.06.004.
- Malik K, Ahmad M, Zhang G, Rashid N, Zafar M, Sultana S, Shah SN. 2018. Traditional plant based medicines used to treat musculoskeletal disorders in Northern Pakistan. Eur J Integr Med. 19: 17–64. doi: 10.1016/j.eujim.2018.02.003.
- Manandhar NP. 1995. A survey of medicinal plants of Jajarkot district, Nepal. J Ethnopharmacol. 48(1): 1–6. doi: 10.1016/0378-8741(95)01269-j.
- Manna P, Sinha M, Sil PC. 2009. Prophylactic role of arjunolic acid in response to streptozotocin mediated diabetic renal injury: activation of polyol pathway and oxidative stress responsive signaling cascades. Chem Biol Interact. 181(3): 297–308. doi: 10.1016/j.cbi.2009.08.004.
- Manrique-de-la-Cuba MF, Gamero-Begazo P, Valencia DE, Barazorda-Ccahuana HL, Gómez B. 2019. Theoretical study of the antioxidant capacity of the flavonoids present in the Annona muricata (Soursop) leaves. J Mol Model. 25(7): 200. doi: 10.1007/s00894-019-4083-7.
- Marahel S, Umesha S. 2016. Anti-inflammatory and antinociceptive effect of Pachygone ovata leaves. Pharm Biol. 54(12): 3046–3054. doi: 10.1080/13880209.2016.1203960.
- Marella A, Tanwar OP, Saha R, Ali MR, Srivastava S, Akhter M, Shaquiquzzaman M, Alam MM. 2013. Quinoline: a versatile heterocyclic. Saudi Pharm J. 21(1): 1–12. doi: 10.1016/j.jsps.2012.03.002.
- Mazumder M, Rahman S. 2008. Pharmacological evaluation of Bangladeshi medicinal plants for antioxidant activity. Pharm Biol. 46(10–11): 704–709. doi: 10.1080/13880200802215735.
- Mekky RH, del Mar Contreras M, El-Gindi MR, Abdel-Monem AR, Abdel-Sattar E, Segura-Carretero A. 2015. Profiling of phenolic and other compounds from Egyptian cultivars of chickpea (Cicer arietinum L.) and antioxidant activity: a comparative study. RSC Adv. 5(23): 17751–17767. doi: 10.1039/C4RA13155J.
- Mencherini T, Cau A, Bianco G, Loggia RD, Aquino R, Autore G. 2007. An extract of Apium graveolens var. dulce leaves: structure of the major constituent, apiin, and its anti‐inflammatory properties. J Pharm Pharmacol. 59(6): 891–897. doi: 10.1211/jpp.59.6.0016.
- Miao M, Guo L, Yan X, Wang T, Li Z. 2016. Effects of verbenalin on prostatitis mouse model. Saudi J Biol Sci. 23(1): S148–S157. doi: 10.1016/j.sjbs.2015.10.006.
- Min D, Baek S, Park HW, Lee SA, Moon J, Yang JE, Kim KS, Kim JY, Kang EK. 2016. Prevalence and characteristics of musculoskeletal pain in Korean farmers. Ann Rehabil Med. 40(1): 1–13. doi: 10.5535/arm.2016.40.1.1.
- Ming XF, Rajapakse AG, Carvas JM, Ruffieux J, Yang Z. 2009. Inhibition of S6K1 accounts partially for the anti-inflammatory effects of the arginase inhibitor l-norvaline. BMC Cardiovasc Disord. 9(1): 12. doi: 10.1186/1471-2261-9-12.
- Mishra SK, Tiwari S, Shrivastava A, Srivastava S, Boudh GK, Chourasia SK, Chaturvedi U, Mir SS, Saxena AK, Bhatia G, et al. 2014. Antidyslipidemic effect and antioxidant activity of anthraquinone derivatives from Rheum emodi rhizomes in dyslipidemic rats. J Nat Med. 68(2): 363–371. doi: 10.1007/s11418-013-0810-z.
- Moonjai J. 2017. [Ethnobotany of ethnic group in Mae La Noi District, Mae Hong Son Province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Muangyen N. 2013. [Ethnobotany of Tai Lue and Tai Yuan in Samoeng district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Muhamad AS, Keong CC, Kiew OF, Abdullah MR. 2009. Eurycoma longifolia Jack: medicinal properties and its effect on endurance exercise performance. Asian Exerc Sport Sci J. 6(1): 1–6.
- Murayama M, Ito T, Konno C, Hikino H. 1984. Mechanism of analgesic action of mesaconitine. I. Relationship between analgesic effect and central monoamines or opiate receptors. Eur J Pharmacol. 101(1–2): 29–36. doi: 10.1016/0014-2999(84)90027-x.
- Mutasim DF. 2006. Successful treatment of inflammatory linear verrucous epidermal nevus with tacrolimus and fluocinonide. J Cutan Med Surg. 10(1): 45–47. doi: 10.1007/7140.2006.00004.
- Naik N, Kumar HV, Harini ST. 2011. Synthesis and antioxidant evaluation of novel indole-3-acetic acid analogues. Eur J Chem. 2(3): 337–341. doi: 10.5155/eurjchem.2.3.337-341.363.
- Naik N, Sharath V, Kumar HV. 2012. Novel indole-2-carboxylic acid analogues: synthesis and a new light in to their antioxidant potentials. Eur J Chem. 3(2): 214–219. doi: 10.5155/eurjchem.3.2.214-219.588.
- Nam SY, Kim HY, Yoou MS, Kim AH, Park BJ, Jeong HJ, Kim HM. 2015. Anti-inflammatory effects of isoacteoside from Abeliophyllum distichum. Immunopharmacol Immunotoxicol. 37(3): 258–264. doi: 10.3109/08923973.2015.1026604.
- Nardi M, Bonacci S, De Luca G, Maiuolo J, Oliverio M, Sindona G, Procopio A. 2014. Biomimetic synthesis and antioxidant evaluation of 3, 4-DHPEA-EDA [2-(3, 4-hydroxyphenyl) ethyl (3S, 4E)-4-formyl-3-(2-oxoethyl) hex-4-enoate]. Food Chem. 162: 89–93. doi: 10.1016/j.foodchem.2014.04.015.
- Natsume M, Osakabe N, Oyama M, Sasaki M, Baba S, Nakamura Y, Osawa T, Terao J. 2003. Structures of (−)-epicatechin glucuronide identified from plasma and urine after oral ingestion of (−)-epicatechin: differences between human and rat. Free Radic Biol Med. 34(7): 840–849. doi: 10.1016/s0891-5849(02)01434-x.
- Natsume M, Osakabe N, Yasuda A, Baba S, Tokunaga T, Kondo K, Osawa T, Terao J. 2004. In vitro antioxidative activity of (−)-epicatechin glucuronide metabolites present in human and rat plasma. Free Radic Res. 38(12): 1341–1348. doi: 10.1080/10715760400022087.
- Nessa F, Ismail Z, Mohamed N, Haris MRHM. 2004. Free radical-scavenging activity of organic extracts and of pure flavonoids of Blumea balsamifera DC leaves. Food Chem. 88(2): 243–252. doi: 10.1016/j.foodchem.2004.01.041.
- Nguyen XMA, Bun SS, Ollivier E, Dang TPT. 2020b. Ethnobotanical study of medicinal plants used by K’Ho-Cil people for treatment of diarrhea in Lam Dong province, Vietnam. J Herb Med. 19: 1–41.
- Nguyen TXT, Dang DL, Ngo VQ, Trinh TC, Trinh QN, Do TD, Thanh TTT. 2020a. Anti-inflammatory activity of a new compound from Vernonia amygdalina. Nat Prod Res.1–6.
- Nićiforović N, Abramovič H. 2014. Sinapic acid and its derivatives: natural sources and bioactivity. Compr Rev Food Sci Food Saf. 13(1): 34–51. doi: 10.1111/1541-4337.12041.
- Niu X, Xing W, Li W, Fan T, Hu H, Li Y. 2012. Isofraxidin exhibited anti-inflammatory effects in vivo and inhibited TNF-α production in LPS-induced mouse peritoneal macrophages in vitro via the MAPK pathway. Int Immunopharmacol. 14(2): 164–171. doi: 10.1016/j.intimp.2012.06.022.
- Nunes CDR, Barreto Arantes M, Menezes de Faria Pereira S, Leandro da Cruz L, de Souza Passos M, Pereira de Moraes L, Vieira IJC, Barros de Oliveira D. 2020. Plants as sources of anti-inflammatory agents. Molecules. 25(16): 3726. doi: 10.3390/molecules25163726.
- Oberlies NH, Kim NC, Brine DR, Collins BJ, Handy RW, Sparacino CM, Wani MC, Wall ME. 2004. Analysis of herbal teas made from the leaves of comfrey (Symphytum officinale): reduction of N-oxides results in order of magnitude increases in the measurable concentration of pyrrolizidine alkaloids. Public Health Nutr. 7(7): 919–924. doi: 10.1079/phn2004624.
- Oghenejobo M, Bethel O. 2017. Antibacterial evaluation, phytochemical screening and ascorbic acid assay of turmeric (Curcuma longa). MOJ Bioequiv Availab. 4: 00063.
- Oh IH, Yoon SJ, Seo HY, Kim EJ, Kim Y. 2011. The economic burden of musculoskeletal disease in Korea: a cross sectional study. BMC Musculoskel Disord. 12(1): 1–9.
- Okuyama E, Fujimori S, Yamazaki M, Deyama T. 1998. Pharmacologically active components of viticis fructus (Vitex rotundifolia). II. The component having analgesic effects. Chem Pharm Bull (Tokyo). 46(4): 655–662. doi: 10.1248/cpb.46.655.
- Omidi-Ardali H, Lorigooini Z, Soltani A, Balali-Dehkordi S, Amini-Khoei H. 2019. Inflammatory responses bridge comorbid cardiac disorder in experimental model of IBD induced by DSS: protective effect of the trigonelline. Inflammopharmacology. 27(6): 1265–1273. doi: 10.1007/s10787-019-00581-w.
- Omran A, Reza G, Alizadeh S, Yahya R, Pouria SD. 2015. Prevalence of musculoskeletal disorders among farmers in eastern Azerbaijan, Iran. Indian J Sci Technol. 8(28): 1–6.
- Ozaki Y. 1990. Antiinflammatory effect of Curcuma xanthorrhiza ROXB. and its active principles. Chem Pharm Bull (Tokyo). 38(4): 1045–1048. doi: 10.1248/cpb.38.1045.
- Pan T, Wu D, Cai N, Chen R, Shi X, Li B, Pan J. 2017. Alpha-Mangostin protects rat articular chondrocytes against IL-1β-induced inflammation and slows the progression of osteoarthritis in a rat model. Int Immunopharmacol. 52: 34–43. doi: 10.1016/j.intimp.2017.08.010.
- Pandey A, Bani S, Satti NK, Gupta BD, Suri KA. 2012. Anti-arthritic activity of agnuside mediated through the down-regulation of inflammatory mediators and cytokines. Inflamm Res. 61(4): 293–304. doi: 10.1007/s00011-011-0410-x.
- Pang Y, Wang D, Fan Z, Chen X, Yu F, Hu X, Wang K, Yuan L. 2014a. Blumea balsamifera—A phytochemical and pharmacological review. Molecules. 19(7): 9453–9477. doi: 10.3390/molecules19079453.
- Pang Y, Wang D, Hu X, Wang H, Fu W, Fan Z, Chen X, Yu F. 2014b. Effect of volatile oil from Blumea balsamifera (L.) DC. leaves on wound healing in mice. J Tradit Chin Med. 34(6): 716–724. doi: 10.1016/s0254-6272(15)30087-x.
- Pang Z, Zhi-Yan Z, Wang W, Ma Y, Feng-Ju N, Zhang X, Han C. 2015. The advances in research on the pharmacological effects of Fructus Ligustri Lucidi. Biomed Res Int. 2015:281873–281875. doi: 10.1155/2015/281873.
- Panya A, Pundith H, Thongyim S, Kaewkod T, Chitov T, Bovonsombut S, Tragoolpua Y. 2020. Antibiotic-antiapoptotic dual function of Clinacanthus nutans (Burm. F.) Lindau leaf extracts against bovine mastitis. Antibiotics. 9(7): 429. doi: 10.3390/antibiotics9070429.
- Panyadee P. 2012. Plant diversity in homegardens of Tai Yai communities in Pang Mapha district, Mae Hong Son province. Chiang Mai, Thailand: Chiang Mai University.
- Park KS. 2020. An overview on anti-inflammatory activities of Aucubin. Int J Herb Med. 8: 45–48.
- Park SB, Jung WH, Kang NS, Park JS, Bae GH, Kim HY, Rhee SD, Kang SK, Ahn JH, Jeong HG, et al. 2013. Anti-diabetic and anti-inflammatory effect of a novel selective 11β-HSD1 inhibitor in the diet-induced obese mice. Eur J Pharmacol. 721(1–3): 70–79. doi: 10.1016/j.ejphar.2013.09.052.
- Park S, Nhiem NX, Van Kiem P, Van Minh C, Tai BH, Kim N, Yoo HH, Song JH, Ko HJ, Kim SH. 2014. Five new quassinoids and cytotoxic constituents from the roots of Eurycoma longifolia. Bioorg Med Chem Lett. 24(16): 3835–3840. doi: 10.1016/j.bmcl.2014.06.058.
- Park KD, Park YS, Cho SJ, Sun WS, Kim SH, Jung DH, Kim JH. 2004. Antimicrobial activity of 3-O-acyl-(-)-epicatechin and 3-O-acyl-(+)-catechin derivatives. Planta Med. 70(3): 272–276. doi: 10.1055/s-2004-818923.
- Patel SS, Savjani JK. 2015. Systematic review of plant steroids as potential antiinflammatory agents: current status and future perspectives. J Phytopharmacol. 4(2): 121–125. doi: 10.31254/phyto.2015.4212.
- Pawar RS, Bhutani K. 2004. Madhucosides A and B, protobassic acid glycosides from Madhuca indica with inhibitory A activity on free radical release from phagocytes. J Nat Prod. 67(4): 668–671. doi: 10.1021/np0301792.
- Pei K, Ou J, Huang J, Ou S. 2016. p‐Coumaric acid and its conjugates: dietary sources, pharmacokinetic properties and biological activities. J Sci Food Agric. 96(9): 2952–2962. doi: 10.1002/jsfa.7578.
- Peng H, Deng Z, Chen X, Sun Y, Zhang B, Li H. 2018. Major chemical constituents and antioxidant activities of different extracts from the peduncles of Hovenia acerba Lindl. Int J Food Prop. 21(1): 2135–2155. doi: 10.1080/10942912.2018.1497059.
- Peters RR, Farias MR, Ribeiro-do-Valle RM. 1997. Anti-inflammatory and analgesic effects of cucurbitacins from Wilbrandia ebracteata. Planta Med. 63(6): 525–528. doi: 10.1055/s-2006-957755.
- Petricevich VL, Abarca-Vargas R. 2019. Allamanda cathartica: a review of the phytochemistry, pharmacology, toxicology, and biotechnology. Molecules. 24(7): 1238. doi: 10.3390/molecules24071238.
- Petronzi C, Filosa R, Peduto A, Monti MC, Margarucci L, Massa A, Ercolino SF, Bizzarro V, Parente L, Riccio R, et al. 2011. Structure-based design, synthesis and preliminary anti-inflammatory activity of bolinaquinone analogues. Eur J Med Chem. 46(2): 488–496. doi: 10.1016/j.ejmech.2010.11.028.
- Phumthum M, Balslev H, Kantasrila R, Kaewsangsai S, Inta A. 2020. Ethnomedicinal plant knowledge of the Karen in Thailand. Plants. 9(7): 813. doi: 10.3390/plants9070813.
- Phumthum M, Srithi K, Inta A, Junsongduang A, Tangjitman K, Pongamornkul W, Trisonthi C, Balslev H. 2018. Ethnomedicinal plant diversity in Thailand. J Ethnopharmacol. 214: 90–98. doi: 10.1016/j.jep.2017.12.003.
- Pinto NB, Morais TC, Carvalho K, Silva CR, Andrade GMD, Brito GADC, Veras ML, Pessoa ODL, Rao VS, Santos FA. 2010. Topical anti-inflammatory potential of physalin E from Physalis angulata on experimental dermatitis in mice. Phytomedicine. 17(10): 740–743. doi: 10.1016/j.phymed.2010.01.006.
- Pipitkul S. 2001. Medicinal plant utilization for the living of Hill Tribes at Doi Musoe, Tak Province. Bangkok, Thailand: Mahidol University.
- Pollini L, Tringaniello C, Ianni F, Blasi F, Manes J, Cossignani L. 2020. Impact of ultrasound extraction parameters on the antioxidant properties of Moringa oleifera leaves. Antioxidants. 9(4): 277. doi: 10.3390/antiox9040277.
- Pongamornkul W. 2003. An ethnobotanical study of the Karen at Ban Yang Pu Toh and Ban Yang Thung Pong, Chiang Dao district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Pongamornkul W. 2006a. An ethnobotanical study of Lau in Royal Project Area, Mae Hong Son province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Pongamornkul W. 2006b. Report on the study of plant utilization of various communities un the upper Sa River Valley. Thailand: The Botanical Garden Organization, Ministry of Natural Resources and Environment. Thai.
- Pongamornkul W. 2010. Survey and collection of medicinal plants and traditional plants of the ethnic groups in Northern of Thailand: annual performance report 2010. Thailand: The Botanical Garden Organization, Ministry of Natural Resources and Environment. Thai.
- Pongamornkul W. 2017. Northern Thailand ethnobotanical index. Thailand: wanida Karnpim Limited Partmership. Thai.
- Pongamornkul W. 2009. Annual report of the project collecting and exhibiting local plants and Lanna herbs. Annual report in 2010. Garden QSB, editor. Chiang Mai, Thailand: Queen Sirikit Botanical Garden. Thai.
- Pongamornkul W, Muangyen N. 2013. Ethnobotany of Karen in Sop moei district, Mae Hong Son province. Annual report in 2013. The ethbobotany of ethnic groups in Nortern of Thailand. Chiang Mai, Thailand: queen Sirikit Botanical Garden. Thai
- Pongamornkul W, Muangyen N. 2012. Ethnobotany of Tai Yai in Khun Yuam district, Mae Hong Son province. Annual report in 2013. In: Garden QSB, editor. The ethbobotany of ethnic groups in Northern of Thailand. Thailand: The Botanical Garden Organization, Ministry of Natural Resources and Environment. Thai.
- Pongsattayapipat R. 1992. Ethnobotanical study of the Hmong Khao, Chang Khian village, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Ponpim Y. 1996. Ethnobotany of the hill tribes in Kaenoy’s and Nongkheuw’s Royal Project in Chiang Mai. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Prance GT, Balée W, Boom BM, Carneiro RL. 1987. Quantitative ethnobotany and the case for conservation in Ammonia. Conserv Biol. 1(4): 296–310. doi: 10.1111/j.1523-1739.1987.tb00050.x.
- Puling W. 2001. Ethnobotany of Karen for studying medicinal plants at Angka Noi and Mae Klangluang villages, Chomthong district, Chiang Mai. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Puntumetakul R, Siritaratiwat W, Boonprakob Y, Eungpinichpong W, Puntumetakul M. 2011. Prevalence of musculoskeletal disorders in farmers: case study in Sila, Muang Khon Kaen, Khon Kaen province. J Med Tech Phys Ther. 23: 297–303.
- Qais N, Etu SF. 2020. Antioxidant activity of lumichrome and its reduced form, 5,10-dihydro-7,8-dimethyl Alloxazine. J Pharm Biol Sci. 15: 50–53.
- Qin X, Chen J, Zhang G, Li C, Zhu J, Xue H, Li J, Guan T, Zheng H, Liu Y, et al. 2020. Hydroxysafflor yellow A exerts anti-inflammatory effects mediated by SIRT1 in lipopolysaccharide-induced microglia activation. Front Pharmacol. 11: 1315. doi: 10.3389/fphar.2020.01315.
- Qnais E, Raad D, Bseiso Y. 2014. Analgesic and anti-inflammatory effects of an extract and flavonoids from Artemisia herba-alba and their mechanisms of action. Neurophysiology. 46(3): 238–246. doi: 10.1007/s11062-014-9435-4.
- Rahimi A, Razmkhah K, Mehrnia M, Mohamadnia A, Sahebjamee H, Salehi S, Asl EA, Tahmasebi H, Shandiz SAS, Davouodbeglou F, et al. 2016. Molecular docking and binding study of harpagoside and harpagide as novel anti-inflammatory and anti-analgesic compound from Harpagophytum procumbens based on their interactions with COX-2 enzyme. Asian Pac J Trop Dis. 6(3): 227–231. doi: 10.1016/S2222-1808(15)61019-2.
- Rahman R, Sabahi J, Ghaffar A, Nadeem F, Umar A. 2019. Phytochemical, morphological, botanical and pharmacological aspects of a medicinal plant: kalanchoe pinnata – a review article. Int J Chem Biochem Sci. 16: 5–10.
- Rajbhandari KR. 2001. Ethnobotany of Nepal. English: Ethnobotanical Society of Nepal.
- Ramezani M, Nasri S, Yassa N. 2009. Antinociceptive and anti-inflammatory effects of isolated fractions from Apium graveolens seeds in mice. Pharm Biol. 47(8): 740–743. doi: 10.1080/13880200902939283.
- Rastogi S, Pandey MM, Rawat AKS. 2015. Medicinal plants of the genus Betula—traditional uses and a phytochemical–pharmacological review. J Ethnopharmacol. 159: 62–83. doi: 10.1016/j.jep.2014.11.010.
- Riaz A, Rasul A, Hussain G, Zahoor MK, Jabeen F, Subhani Z, Younis T, Ali M, Sarfraz I, Selamoglu Z. 2018. Astragalin: a bioactive phytochemical with potential therapeutic activities. Adv Pharmacol Sci. 2018: 9794625. doi: 10.1155/2018/9794625.
- Rice-Evans CA, Miller NJ, Bolwell PG, Bramley PM, Pridham JB. 1995. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic Res. 22(4): 375–383. doi: 10.3109/10715769509145649.
- Rimpelová S, Jurášek M, Peterková L, Bejček J, Spiwok V, Majdl M, Jirásko M, Buděšínský M, Harmatha J, Kmoníčková E, et al. 2019. Archangelolide: a sesquiterpene lactone with immunobiological potential from Laserpitium archangelica. Beilstein J Org Chem. 15: 1933–1944. doi: 10.3762/bjoc.15.189.
- Rosecrance J, Rodgers G, Merlino L. 2006. Low back pain and musculoskeletal symptoms among Kansas farmers. Am J Ind Med. 49(7): 547–556. doi: 10.1002/ajim.20324.
- Royal Forest Department [RFD]. 2022. Forest Land Manangment Office RFD, editor. Executive summary 2022: forest area information project. Forest Land Manangment Office, Royal Forest Department: ministry of Natural Resources and Environment.
- Ruiz-Cruz S, Chaparro-Hernández S, Hernández-Ruiz KL, Cira-Chávez LA, Estrada-Alvarado MI, Ortega LEG, Mata M. 2017. Flavonoids: important biocompounds in food. In: Justino G, editor. Flavonoids - from biosynthesis to human health. InTechOpen, p. 353–369.
- Saeed O, Arif U, Saqib MU, Sidiqque A, Khalid H, Shahzad A. 2021. A descriptive study of prevalence of musculoskeletal illnesses among farmers in Faisalabad, Pakistan. J Nov Physiother. 8(1): 001–004.
- Sakamoto J, Umeda Y, Rakumitsu K, Sumimoto M, Ishikawa H. 2020. Total syntheses of (−)‐strictosidine and related indole alkaloid glycosides. Angew Chem Int Ed Engl. 59(32): 13414–13422. doi: 10.1002/anie.202005748.
- Salatino A, Salatino MLF, Negri G. 2007. Traditional uses, chemistry and pharmacology of Croton species (Euphorbiaceae). J Braz Chem Soc. 18(1): 11–33. doi: 10.1590/S0103-50532007000100002.
- Saleem H, Htar TT, Naidu R, Zengin G, Locatelli M, Tartaglia A, Zainal Abidin SA, Ahemad N. 2020. Phytochemical composition and enzyme inhibition studies of Buxus papillosa CK Schneid. Processes. 8(7): 757. doi: 10.3390/pr8070757.
- Salehi B, Albayrak S, Antolak H, Kręgiel D, Pawlikowska E, Sharifi-Rad M, Uprety Y, Tsouh Fokou PV, Yousef Z, Amiruddin Zakaria Z, et al. 2018. Aloe genus plants: from farm to food applications and phytopharmacotherapy. Int J Mol Sci. 19(9):2843. doi: 10.3390/ijms19092843.
- Salehi B, Sharopov F, Boyunegmez Tumer T, Ozleyen A, Rodríguez-Pérez C, Ezzat SM, Azzini E, Hosseinabadi T, Butnariu M, Sarac I, et al. 2019. Symphytum species: a comprehensive review on chemical composition, food applications and phytopharmacology. Molecules. 24(12): 2272. doi: 10.3390/molecules24122272.
- Samoisy AK, Mahomoodally F. 2016. Ethnopharmacological appraisal of culturally important medicinal plants and polyherbal formulas used against communicable diseases in Rodrigues Island. J Ethnopharmacol. 194: 803–818. doi: 10.1016/j.jep.2016.10.041.
- Samu Z, Nyiredy S, Baitz‐Gács E, Varga Z, Kurtán T, Dinya Z, Antus S. 2004. Structure elucidation and antioxidant activity of (−)‐isosilandrin isolated from Silybum marianum L. Chem Biodivers. 1(11): 1668–1677. doi: 10.1002/cbdv.200490125.
- Sancho R, Lucena C, Macho A, Calzado MA, Blanco‐Molina M, Minassi A, Appendino G, Muñoz E. 2002. Immunosuppressive activity of capsaicinoids: capsiate derived from sweet peppers inhibits NF‐κB activation and is a potent antiinflammatory compound in vivo. Eur J Immunol. 32(6): 1753–1763. doi: 10.1002/1521-4141(200206)32:6<1753::AID-IMMU1753>3.0.CO;2-2.
- Santasombat Y. 2004. Karen cultural capital and the political economy of symbolic power. Asian Ethn. 5: 105–120.
- Santos EN, Lima J, Noldin VF, Cechinel-Filho V, Rao VS, Lima EF, Schmeda-Hirschmann G, Sousa PT, Jr, Martins DT. 2011. Anti-inflammatory, antinociceptive, and antipyretic effects of methanol extract of Cariniana rubra stem bark in animal models. An Acad Bras Cienc. 83(2): 557–566. doi: 10.1590/s0001-37652011005000006.
- Sarı A, Şahin H, Özsoy N, Çelik BÖ. 2019. Phenolic compounds and in vitro antioxidant, anti-inflammatory, antimicrobial activities of Scorzonera hieraciifolia Hayek roots. S Afr J Bot. 125: 116–119. doi: 10.1016/j.sajb.2019.07.009.
- Sasi S, Anjum N, Tripathi Y. 2018. Ethnomedicinal, phytochemical and pharmacological aspects of Flacourtia jangomas: a review. Int J Pharm Pharm Sci. 10(3): 9–15. doi: 10.22159/ijpps.2018v10i3.23998.
- Savtekin G, Serakinci N, Erzik C, Cetinel S, Sehirli A. 2018. Effects of circadian rhythm hormones melatonin and 5-methoxytryptophol on COXs, Raf-1 and STAT3. International J of Pharmacology. 14(6): 787–795. doi: 10.3923/ijp.2018.787.795.
- Sayed N, Khurana A, Godugu C. 2019. Pharmaceutical perspective on the translational hurdles of phytoconstituents and strategies to overcome. J Drug Deliv Sci Technol. 53: 101201. doi: 10.1016/j.jddst.2019.101201.
- Schliesinger J. 2000. Ethnic groups of Thailand: non-Tai-speaking peoples. Bangkok, Thailand: White Lotus Co., Ltd.
- Serafini M, Peluso I, Raguzzini A. 2010. Flavonoids as anti-inflammatory agents. Proc Nutr Soc. 69(3): 273–278. doi: 10.1017/S002966511000162X.
- Servili M, Esposto S, Fabiani R, Urbani S, Taticchi A, Mariucci F, Selvaggini R, Montedoro G. 2009. Phenolic compounds in olive oil: antioxidant, health and organoleptic activities according to their chemical structure. Inflammopharmacology. 17(2): 76–84. doi: 10.1007/s10787-008-8014-y.
- Shan M-Q, Wang T-J, Jiang Y-L, Yu S, Yan H, Zhang L, Wu Q-N, Geng T, Huang W-Z, Wang Z-Z, et al. 2019. Comparative analysis of sixteen active compounds and antioxidant and anti-influenza properties of Gardenia jasminoides fruits at different times and application to the determination of the appropriate harvest period with hierarchical cluster analysis. J Ethnopharmacol. 233: 169–178. doi: 10.1016/j.jep.2019.01.004.
- Shanawany MA, Sayed HM, Ibrahim SR, Fayed MA. 2014. Chemical constituents, anti-inflammatory, and antioxidant activities of Anisotes trisulcus. Bull Fac Pharm Cairo Univ. 52(1): 9–14. doi: 10.1016/j.bfopcu.2014.02.004.
- Shankaranarayan D, Gopalakrishnan C, Kameswaran L. 1979. Pharmacological profile of mangostin and its derivatives. Arch Int Pharmacodyn Ther. 239(2): 257–269.
- Sharif M, Najafizadeh P, Asgarpanah J, Mousavi Z. 2020. In vivo analgesic and anti-inflammatory effects of the essential oil from Tanacetum balsamita L. Braz J Pharm Sci. 56: e18357. doi: 10.1590/s2175-97902019000418357.
- Sharma RA, Euden SA, Platton SL, Cooke DN, Shafayat A, Hewitt HR, Marczylo TH, Morgan B, Hemingway D, Plummer SM, et al. 2004. Phase I clinical trial of oral curcumin: biomarkers of systemic activity and compliance. Clin Cancer Res. 10(20): 6847–6854. doi: 10.1158/1078-0432.CCR-04-0744.
- Sharma V, Rao LJM. 2009. A thought on the biological activities of black tea. Crit Rev Food Sci Nutr. 49(5): 379–404. doi: 10.1080/10408390802068066.
- Sheng-Ji P. 2001. Ethnobotanical approaches of traditional medicine studies: some experiences from Asia. Pharmaceutical Biol. 39(1): 74–79. doi: 10.1076/phbi.39.7.74.5869.
- Shi C, Li H, Yang Y, Hou L. 2015. Anti-inflammatory and immunoregulatory functions of artemisinin and its derivatives. Mediators Inflamm. 2015: 435713–435717. doi: 10.1155/2015/435713.
- Shin JS, Yun KJ, Chung KS, Seo KH, Park HJ, Cho YW, Baek NI, Jang D, Lee KT. 2013. Monotropein isolated from the roots of Morinda officinalis ameliorates proinflammatory mediators in RAW 264.7 macrophages and dextran sulfate sodium (DSS)-induced colitis via NF-κB inactivation. Food Chem Toxicol. 53: 263–271. doi: 10.1016/j.fct.2012.12.013.
- Shintaku K, Uchida K, Suzuki Y, Zhou Y, Fushiki T, Watanabe T, Yazawa S, Tominaga M. 2012. Activation of transient receptor potential A1 by a non‐pungent capsaicin‐like compound, capsiate. Br J Pharmacol. 165(5): 1476–1486. doi: 10.1111/j.1476-5381.2011.01634.x.
- Shirota O, Oribello JM, Sekita S, Satake M. 2011. Sesquiterpenes from Blumea balsamifera. J Nat Prod. 74(3): 470–476. doi: 10.1021/np100646n.
- Shrestha S, Lee DY, Park JH, Cho JG, Lee DS, Li B, Kim YC, Kim GS, Bang MH, Baek NI. 2013. Phenolic components from Rhus parviflora fruits and their inhibitory effects on lipopolysaccharide-induced nitric oxide production in RAW 264.7 macrophages. Nat Prod Res. 27(23): 2244–2247. doi: 10.1080/14786419.2013.814050.
- Siebert DA, Paganelli CJ, Queiroz GS, Alberton MD. 2020. Anti-inflammatory activity of the epicuticular wax and its isolated compounds catechin and gallocatechin from Eugenia brasiliensis Lam.(Myrtaceae) leaves. Nat Prod Res. 35(22): 4720–4723. doi: 10.1080/14786419.2019.1710707.
- Simamora A, Santoso AW, Timotius KH, Rahayu I. 2020. Antioxidant activity, enzyme inhibition potentials, and phytochemical profiling of Premna serratifolia L. leaf extracts. Int J Food Sci. 2020: 3436940–3436911. doi: 10.1155/2020/3436940.
- Singh V, Kapahi B, Srivastava T. 1996. Medicinal herbs of Ladakh especially used in home remedies. Fitoterapia. 67: 38–48.
- Snafi A. 2016. The contents and pharmacological importance of Corchorus capsularis – a review. IOSR J Pharm. 6: 58–63.
- Sombatsawat E, Luangwilai T, Ong-Artborirak P, Siriwong W. 2019. Musculoskeletal disorders among rice farmers in Phimai district, Nakhon Ratchasima province, Thailand. J Health Res. 33: 2586–940X.
- Song CH, Kim N, Kim DH, Lee HN, Surh YJ. 2019. 17-β estradiol exerts anti-inflammatory effects through activation of Nrf2 in mouse embryonic fibroblasts. PLoS One. 14(8): e0221650. doi: 10.1371/journal.pone.0221650.
- Songsangchun A. 2015. Plants usages of Khon Muang and Lawa in Phu Fah Subdistrict, Bo Klua district, Nan province. Chiang Mai, Thailand: Chiang Mai University.
- Song L, Wang Q, Chai Z, Zhang C, Wang J, Ma J, Guo J, Yu J, Xiao B, Han G, et al. 2020. Anhydrosafflor Yellow B alleviates brain injury of acute permanent cerebral ischemia in rats by anti-inflammatory mechanism. All Life. 13(1): 201–212. doi: 10.1080/26895293.2020.1750492.
- Srithi K. 2012. Comparative ethnobotany in Nan province, Thailand. Chiang Mai, Thailand: Chiang Mai University.
- Srithi K, Balslev H, Wangpakapattanawong P, Srisanga P, Trisonthi C. 2009. Medicinal plant knowledge and its erosion among the Mien (Yao) in northern Thailand. J Ethnopharmacol. 123(2): 335–342. doi: 10.1016/j.jep.2009.02.035.
- Ștefănescu BE, Szabo K, Mocan A, Crişan G. 2019. Phenolic compounds from five Ericaceae species leaves and their related bioavailability and health benefits. Molecules. 24(11): 2046. doi: 10.3390/molecules24112046.
- Su S, Wink M. 2015. Natural lignans from Arctium lappa as antiaging agents in Caenorhabditis elegans. Phytochemistry. 117: 340–350. doi: 10.1016/j.phytochem.2015.06.021.
- Subhan F, Karim N, Ibrar M. 2007. Anti-inflammatory activity of methanolic and aqueous extracts of Valeriana wallichii DC rhizome. Pak J Plant Sci. 13: 103–108.
- Sukkho T. 2008. A Survey of medicinal plants used by Karen People at Ban Chan and Chaem Luang subdistricts, Mae Chaem district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Sun J, Mustafi R, Cerda S, Chumsangsri A, Xia YR, Li YC, Bissonnette M. 2008. Lithocholic acid down-regulation of NF-κB activity through vitamin D receptor in colonic cancer cells. J Steroid Biochem Mol Biol. 111(1–2): 37–40. doi: 10.1016/j.jsbmb.2008.01.003.
- Sun N, Qiu Y, Zhu Y, Liu J, Zhang H, Zhang Q, Zhang M, Zheng G, Zhang C, Yao G. 2019. Rhodomicranosides A − I, analgesic diterpene glucosides with diverse carbon skeletons from Rhododendron micranthum. Phytochemistry. 158: 1–12. doi: 10.1016/j.phytochem.2018.10.017.
- Sun Z, Yang L, Zhao L, Cui R, Yang W. 2020. Neuropharmacological effects of Mesaconitine: evidence from molecular and cellular basis of neural circuit. Neural Plast. 2020: 8814531–8814510. doi: 10.1155/2020/8814531.
- Supawimolpan W. 2011. Ethnobotany of Khamu in Ban Huai Sa Taeng, Thung Chang district, Nan province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Sur R, Nigam A, Grote D, Liebel F, Southall MD. 2008. Avenanthramides, polyphenols from oats, exhibit anti-inflammatory and anti-itch activity. Arch Dermatol Res. 300(10): 569–574. doi: 10.1007/s00403-008-0858-x.
- Sur TK, Pandit S, Battacharyya D, Kumar CA, Lakshmi SM, Chatttopadhyay D, Mandal SC. 2002. Studies on the antiinflammatory activity of Betula alnoides bark. Phytother Res. 16(7):669–671. doi: 10.1002/ptr.942.
- Szekalska M, Sosnowska K, Tomczykowa M, Winnicka K, Kasacka I, Tomczyk M. 2020. In vivo anti-inflammatory and anti-allergic activities of cynaroside evaluated by using hydrogel formulations. Biomed Pharmacother. 121: 109681. doi: 10.1016/j.biopha.2019.109681.
- Tada M, Matsumoto R, Yamaguchi H, Chiba K. 1996. Novel antioxidants isolated from Perilla frutescens Britton var. crispa (Thunb.)Biosci, Biotechnol, Biochem. 60: 1093–1095.
- Tai A, Sawano T, Yazama F. 2011. Antioxidant properties of ethyl vanillin in vitro and in vivo. Biosci Biotechnol Biochem. 75(12): 2346–2350. doi: 10.1271/bbb.110524.
- Tangjitman K. 2014. Vulnerability prediction of medicinal plants used by Karen people in Chiang Mai province to climatic change using species distribution model (SDM). Chiang Mai, Thailand: Chiang Mai University.
- Tangjitman K. 2017. Ethnobotany of the Karen at Huay Nam Nak village, Tanaosri subdistrict, Suanphueng district, Ratchaburi province. Thai J Bot. 9: 253–272.
- Tangjitman K, Trisonthi C, Wongsawad C, Jitaree S, Svenning JC. 2015. Potential impact of climatic change on medicinal plants used in the Karen women’s health care in northern Thailand. Songklanakarin J Sci Technol. 37: 369–379.
- Teiten MH, Mack F, Debbab A, Aly AH, Dicato M, Proksch P, Diederich M. 2013. Anticancer effect of altersolanol A, a metabolite produced by the endophytic fungus Stemphylium globuliferum, mediated by its pro-apoptotic and anti-invasive potential via the inhibition of NF-κB activity. Bioorg Med Chem. 21(13): 3850–3858. doi: 10.1016/j.bmc.2013.04.024.
- Teponno RB, Kusari S, Spiteller M. 2016. Recent advances in research on lignans and neolignans. Nat Prod Rep. 33(9): 1044–1092. doi: 10.1039/c6np00021e.
- Tong Y, Liu S, Gong R, Zhong L, Duan X, Zhu Y. 2019. Ethyl vanillin protects against kidney injury in diabetic nephropathy by inhibiting oxidative stress and apoptosis. Oxid Med Cell Longev. 2019: 2129350–2129312. doi: 10.1155/2019/2129350.
- Torun A, Yilmaz M, Ozkan N, Ustun B, Koksal E, Kaya C. 2017. Sedative-analgesic activity of remifentanil and effects of preoperative anxiety on perceived pain in outpatient mandibular third molar surgery. Int J Oral Maxillofac Surg. 46(3): 379–384. doi: 10.1016/j.ijom.2016.11.005.
- Tovaranonte J. 1998. Ethnobotany study of the Tai Lue, Hmong and Yao in some areas of Nan province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Trahan RA, Byron CR, Dahlgren LA, Pleasant RS, Werre SR. 2018. In vitro effects of three equimolar concentrations of methylprednisolone acetate, triamcinolone acetonide, and isoflupredone acetate on equine articular tissue cocultures in an inflammatory environment. Am J Vet Res. 79(9): 933–940. doi: 10.2460/ajvr.79.9.933.
- Trisonthi C, Trisonthi P. 2009. Ethnobotanical study in Thailand, a case study in Khun Yuam district Maehongson province. Thai J Bot. 1: 1–23.
- Trotter RT, Logan MH. 1986. Informant consensus: a new approach for identifying potentially effective medicinal plants. Bredfort Hill, New York: Redgrave Publishing Company.
- Tu J, Guo Y, Hong W, Fang Y, Han D, Zhang P, Wang X, Körner H, Wei W. 2019. The regulatory effects of paeoniflorin and its derivative paeoniflorin-6′-O-benzene sulfonate CP-25 on inflammation and immune diseases. Front Pharmacol. 10: 57. doi: 10.3389/fphar.2019.00057.
- Udompanid K. 2012. Ethnobotany of Tai Yai in Naisoi village, Mueng district, Mae Hong Son province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Ullah H, Khan A, Baig MW, Ullah N, Ahmed N, Tipu MK, Ali H, Khan S. 2020. Poncirin attenuates CCL4-induced liver injury through inhibition of oxidative stress and inflammatory cytokines in mice. BMC Complement Med Ther. 20(1): 115. doi: 10.1186/s12906-020-02906-7.
- Ványolós A, Muszyńska B, Chuluunbaatar B, Gdula‐Argasińska J, Kała K, Hohmann J. 2020. Extracts and steroids from the edible mushroom Hypholoma lateritium exhibit anti‐inflammatory properties by inhibition of COX‐2 and activation of Nrf2. Chem Biodivers. 17(9): e2000391. doi: 10.1002/cbdv.202000391.
- Varga E, Barabás C, Tóth A, Boldizsár I, Noszál B, Tóth G. 2019. Phenolic composition, antioxidant and antinociceptive activities of Syringa vulgaris L. bark and leaf extracts. Nat Prod Res. 33(11): 1664–1669. doi: 10.1080/14786419.2018.1425855.
- Varghese C, Ambrose C, JS, Lim Y, Keisaban T. 2013. Antioxidant and anti-inflammatory activity of Eurycoma longifolia Jack, a traditional medicinal plant in Malaysia. Int J Pharm Sci Nanotech. 5: 1875–1878.
- Vyvey M. 2010. Steroids as pain relief adjuvants. Can Fam Physician. 56: 1295–1297.
- Wan W, Xia N, Zhu S, Liu Q, Gao Y. 2020. A novel and high-effective biosynthesis pathway of hesperetin-7-O-glucoside based on the construction of immobilized rhamnosidase reaction platform. Front Bioeng Biotechnol. 8: 608. doi: 10.3389/fbioe.2020.00608.
- Wang W, Chen W, Yang Y, Liu T, Yang H, Xin Z. 2015. New phenolic compounds from Coreopsis tinctoria Nutt. and their antioxidant and angiotensin I-converting enzyme inhibitory activities. J Agric Food Chem. 63(1): 200–207. doi: 10.1021/jf504289g.
- Wang L, Guo T, Guo Y, Xu Y. 2020a. Asiaticoside produces an antidepressant‑like effect in a chronic unpredictable mild stress model of depression in mice, involving reversion of inflammation and the PKA/pCREB/BDNF signaling pathway. Mol Med Rep. 22(3): 2364–2372. doi: 10.3892/mmr.2020.11305.
- Wang YP, Guo Y, Wen PS, Zhao ZZ, Xie J, Yang K, Yang Q, Wang JF, Deng XM. 2020c. Three ingredients of safflower alleviate acute lung injury and inhibit NET release induced by lipopolysaccharide. Mediators Inflamm. 2020: 2720369–2720312.
- Wang J, Li P, Li B, Guo Z, Kennelly EJ, Long C. 2014. Bioactivities of compounds from Elephantopus scaber, an ethnomedicinal plant from southwest China. Evid Based Complement Alternat Med. 2014: 569594–569597. doi: 10.1155/2014/569594.
- Wang J, Liu Y, Xu J, Wang A, Wang Y, Long C. 2013. Studies on medicinal plants of Elephantopus (Compositae). Nat Prod Res Dev. 25: 401–409.
- Wang L, Wang P, Wang D, Tao M, Xu W, Olatunji OJ. 2020b. Anti-inflammatory activities of kukoamine A from the root bark of Lycium chinense Miller. Nat Prod Commun. 15(3): 1934578X2091208. 1934578X20912088. doi: 10.1177/1934578X20912088.
- Wang GH, Zou GX, You XM, Zhang Y, Jiang H, Li F, Li GX. 2017. Tectorigenin and irigenin inhibit lipopolysaccharide-induced nitric oxide synthase expression in murine macrophages. Biomed Res. 28: 5412–5417.
- Ward JBJ, Lajczak NK, Kelly OB, O’Dwyer AM, Giddam AK, Ní Gabhann J, Franco P, Tambuwala MM, Jefferies CA, Keely S, et al. 2017. Ursodeoxycholic acid and lithocholic acid exert anti-inflammatory actions in the colon. Am J Physiol Gastrointest Liver Physiol. 312(6): G550–G558. doi: 10.1152/ajpgi.00256.2016.
- Waripo W. 2012. Ethnobotany of Pok village in Samoeng district, Chiang Mai province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Williams CA, Harborne JB, Geiger H, Hoult JRS. 1999. The flavonoids of Tanacetum parthenium and T. vulgare and their anti-inflammatory properties. Phytochemistry. 51(3): 417–423. doi: 10.1016/s0031-9422(99)00021-7.
- Winjchiyanan P. 1995. Ethnobotany of Karen in Chiang Mai. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Wisuitiprot W, Wisuitiprot V, Kaewtai N, Sreenoulkow P. 2020. Bioactivity study of Tadehagi triquetrum extracts used in Thai folk medicine. J Thai Trad Alt Med. 18: 259–269.
- Wonca International Classification Committee [WICC]. 2005. International classification of primary care. 2nd ed. Oxford: Oxford University Press.
- Wong CW, Seow WK, O’Callaghan JW, Thong YH. 1992. Comparative effects of tetrandrine and berbamine on subcutaneous air pouch inflammation induced by interleukin-1, tumour necrosis factor and platelet-activating factor. Agents Actions. 36(1–2): 112–118. doi: 10.1007/BF01991238.
- Woolf AD, Pfleger B. 2003. Burden of major musculoskeletal conditions. Bull RFK. 81: 646–656.
- World Health Organization [WHO]. 2019. Musculoskeletal conditions. Switzerland: WHO Headquarters in Geneva; [accessed 2019 February 5]. https://www.who.int/news-room/fact-sheets/detail/musculoskeletal-conditions.
- Wu CJ, Cui X, Xiong B, Yang MS, Zhang YX, Liu XM. 2019. Terretonin D1, a new meroterpenoid from marine-derived Aspergillus terreus ML-44. Nat Prod Res. 33(15): 2262–2265. doi: 10.1080/14786419.2018.1493583.
- Wu L, Nie L, Xu Z, Li P, Wang Y, He C, Song J, Yao H. 2020. Comparative and phylogenetic analysis of the complete chloroplast genomes of three Paeonia section Moutan species (Paeoniaceae). Front Genet. 11: 980. doi: 10.3389/fgene.2020.00980.
- Wu X, Song M, Rakariyatham K, Zheng J, Guo S, Tang Z, Zhou S, Xiao H. 2015. Anti-inflammatory effects of 4′-demethylnobiletin, a major metabolite of nobiletin. J Funct Foods. 19(Pt A): 278–287. doi: 10.1016/j.jff.2015.09.035.
- Xiang K, Cai H, Song Z. 2014. Comparison of analgesic effects of remifentanil and fentanyl NCA after pediatric cardiac surgery. J Invest Surg. 27(4): 214–218. doi: 10.3109/08941939.2013.879968.
- Xiang Y, Haixia W, Zenggen L, Yanduo T. 2019. Anti-inflammatory activity of compounds isolated from Swertia mussotii. Nat Prod Res. 33(4): 598–601. doi: 10.1080/14786419.2017.1399385.
- Xu J, Guo Y, Sui T, Wang Q, Zhang Y, Zhang R, Wang M, Guan S, Wang L. 2017b. Molecular mechanisms of anti-oxidant and anti-aging effects induced by convallatoxin in Caenorhabditis elegans. Free Radic Res. 51(5): 529–544. doi: 10.1080/10715762.2017.1331037.
- Xu GK, Qin XY, Wang GK, Xie GY, Li XS, Sun CY, Liu BL, Qin JM. 2017a. Antihyperglycemic, antihyperlipidemic and antioxidant effects of standard ethanol extract of Bombax ceiba leaves in high-fat-diet-and streptozotocin-induced Type 2 diabetic rats. Chin J Nat Med. 15(3): 168–177. doi: 10.1016/S1875-5364(17)30033-X.
- Yamamoto M, Jokura H, Hashizume K, Ominami H, Shibuya Y, Suzuki A, Hase T, Shimotoyodome A. 2013. Hesperidin metabolite hesperetin-7-O-glucuronide, but not hesperetin-3′-O-glucuronide, exerts hypotensive, vasodilatory, and anti-inflammatory activities. Food Funct. 4(9): 1346–1351. doi: 10.1039/c3fo60030k.
- Yan X, Xie G, Zhou J, Milne GW. 2018. Traditional Chinese medicines: molecular structures, Natural sources and applications: molecular structures, Natural Sources and Applications. United Kingdom: Routledge.
- Yang YJ, Yi L, Wang Q, Xie BB, Dong Y, Sha CW. 2017. Anti-inflammatory effects of physalin E from Physalis angulata on lipopolysaccharide-stimulated RAW 264.7 cells through inhibition of NF-κB pathway. Immunopharmacol Immunotoxicol. 39(2): 74–79. doi: 10.1080/08923973.2017.1282514.
- Yaso T. 2000. Ethnobotany of the H′ tin and Lua in Phuphaa subdistrict, Baugleua district, Nan province. Chiang Mai, Thailand: Chiang Mai University. Thai.
- Yimer T, Birru EM, Adugna M, Geta M, Emiru YK. 2020. Evaluation of analgesic and anti-inflammatory activities of 80% methanol root extract of Echinops kebericho M. (Asteraceae). J Inflamm Res. 13:647–658. doi: 10.2147/JIR.S267154.
- Yuan E, Liu B, Ning Z. 2008. Preparation and antioxidant activity of camellianin A from Adinandra nitida leaves. J Food Process Preserv. 32(5): 785–797. doi: 10.1111/j.1745-4549.2008.00214.x.
- Yuan D, Ma B, Yang JY, Xie YY, Wang L, Zhang LJ, Kano Y, Wu CF. 2009. Anti-inflammatory effects of rhynchophylline and isorhynchophylline in mouse N9 microglial cells and the molecular mechanism. Int Immunopharmacol. 9(13–14): 1549–1554. doi: 10.1016/j.intimp.2009.09.010.
- Yue SJ, Tang YP, Wang LY, Tang H, Li SJ, Liu P, Su SL, Duan JA. 2014. Separation and evaluation of antioxidant constituents from Carthamus tinctorius. Zhongguo Zhong Yao Za Zhi. 39(17): 3295–3300.
- Zak A, Siwinska N, Slowikowska M, Borowicz H, Szpot P, Zawadzki M, Niedzwiedz A. 2018. The detection of capsaicin and dihydrocapsaicin in horse serum following long-term local administration. BMC Vet Res. 14(1): 193. doi: 10.1186/s12917-018-1518-9.
- Zhang MH, Feng L, Zhu MM, Gu JF, Wu C, Jia XB. 2013b. Antioxidative and anti-inflammatory activities of paeoniflorin and oxypaeoniflora on AGEs-induced mesangial cell damage. Planta Med. 79(14): 1319–1323. doi: 10.1055/s-0033-1350649.
- Zhang R, Han D, Li Z, Shen C, Zhang Y, Li J, Yan G, Li S, Hu B, Li J, et al. 2018b. Ginkgolide C alleviates myocardial ischemia/reperfusion-induced inflammatory injury via inhibition of CD40-NF-κB pathway. Front Pharmacol. 9: 109. doi: 10.3389/fphar.2018.00109.
- Zhang MR, Jiang K, Yang JL, Shi YP. 2020a. Flavonoids as key bioactive components of Oxytropis falcata Bunge, a traditional anti-inflammatory and analgesic Tibetan medicine. Nat Prod Res. 34(23): 3335–3352. doi: 10.1080/14786419.2019.1574786.
- Zhang D, Li X, Hu Y, Jiang H, Wu Y, Ding Y, Yu K, He H, Xu J, Sun L, et al. 2018a. Tabersonine attenuates lipopolysaccharide-induced acute lung injury via suppressing TRAF6 ubiquitination. Biochem Pharmacol. 154: 183–192. doi: 10.1016/j.bcp.2018.05.004.
- Zhang Y, Ni T, Zhang D, Liu H, Wang J, Sun B. 2020c. Consumption of avenanthramides extracted from oats reduces weight gain, oxidative stress, inflammation and regulates intestinal microflora in high fat diet-induced mice. J Funct Foods. 65: 103774. doi: 10.1016/j.jff.2019.103774.
- Zhang CR, Schutzki RE, Nair MG. 2013a. Antioxidant and anti-inflammatory compounds in the popular landscape plant Berberis thunbergii var. atropurpurea. Nat Prod Commun. 8(2): 1934578X1300800. 1934578X1300800207. doi: 10.1177/1934578X1300800207.
- Zhang L, Wei W. 2020. Anti-inflammatory and immunoregulatory effects of paeoniflorin and total glucosides of paeony. Pharmacol Ther. 207: 107452. doi: 10.1016/j.pharmthera.2019.107452.
- Zhang Y, Yu L, Jin W, Li C, Wang Y, Wan H, Yang J. 2020d. Simultaneous optimization of the ultrasonic extraction method and determination of the antioxidant activities of hydroxysafflor yellow A and anhydrosafflor yellow B from safflower using a response surface methodology. Molecules. 25(5): 1226. doi: 10.3390/molecules25051226.
- Zhang Q, Zhang J-H, He Y-Q, Zhang Q-L, Zhu B, Shen Y, Liu M-Q, Zhu L-L, Xin H-L, Qin L-P, et al. 2020b. Iridoid glycosides from Morinda officinalis How. exert anti-inflammatory and anti-arthritic effects through inactivating MAPK and NF-κB signaling pathways. BMC Complement Med Ther. 20(1): 172. doi: 10.1186/s12906-020-02895-7.
- Zheng QX, Wu HF, Jian G, Nan HJ, Chen SL, Yang JS, Xu XD. 2013. Review of rhubarbs: chemistry and pharmacology. Chin Herb Med. 5: 9–32.
- Zou X, Tan Q, Goh BH, Lee LH, Tan KL, Ser HL. 2020. ‘Sweeter’than its name: anti-inflammatory activities of Stevia rebaudiana. All Life. 13(1): 286–309. doi: 10.1080/26895293.2020.1771434.
- Zou Y, Zhang M, Zhang T, Wu J, Wang J, Liu K, Zhan N. 2018. Antioxidant and anti-inflammatory activities of Cynaroside from Elsholtiza bodinieri. Nat Prod Commun. 13(11): 1934578X1801301. 1934578X1801301122. doi: 10.1177/1934578X1801301122.

