Figures & data
Table 1. Baseline characteristics between included and excluded premature children
Table 2. Vaccination experiences of live attenuated vaccines included in the expanded program of immunization in premature children
Table 3. Vaccination experiences of inactivated vaccines included in the expanded program of immunization in premature children
Figure 1. Vaccination experiences of non-EPI vaccines before and after the assessment by the Vaccination Consultation Clinic (VCC) at the Children’s Hospital of Fudan University. In each panel, the height of colorful bars (left side) and triangles (right side) indicated the number of premature children in each group. Before the assessment, premature children were classified into four groups, demonstrating vaccination status on the left side of each bar, including “vaccinated,” “under RAV (recommended age of vaccination),” “within RAV,” and “beyond RAV.” After the assessment, on the right side of each triangle, premature children were classified as “vaccinated” and “beyond RAV” in the vaccination of PCV13 (panel a), DTaP-IPV/Hib (b), DTaP-Hib (c), and Hib (d); an additional status “within RAV” in EV-71 (e) and RotV (f); and another status “under RAV” in VarV (g). The flows from left bars to right triangles indicted the number of premature children who changed or remained their vaccination status before and after the assessment by VCC.
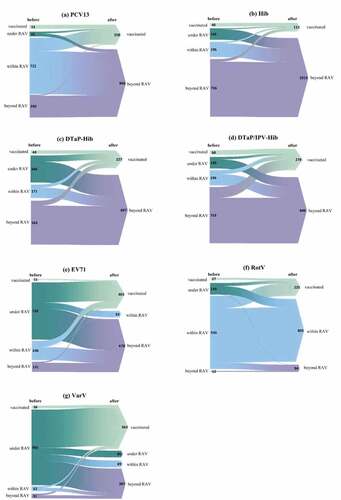
Table 4. Incidence of notifiable vaccine-preventable diseases in included and excluded premature children
Table 5. Incidence of notifiable vaccine-preventable diseases in included children
