ABSTRACT
During the initial half-year of their existence, infants cannot receive the influenza vaccine, yet they face the greatest susceptibility to severe influenza complications. In this study, we seek to determine whether influenza vaccination of maternal and household contacts is associated with a reduced risk of influenza-like illness (ILI) and severe acute respiratory infection (SARI) in infants. This work was prospectively conducted during the influenza season. A total of 206 infants were included in this study. The percentage of infants with only the mother vaccinated is 12.6% (n:26), and the percent of infants with all household contacts vaccinated is 16% (n:33). Among the infants with only the mother vaccinated, the effectiveness of influenza vaccine is estimated as 35.3% for ILI and 41.3% for SARI. Among infants with all household contacts vaccinated, the effectiveness is estimated as 48.9% for ILI and 76.9% for SARI. Based on the results of multivariate logistic regression analysis, all-household vaccination is a protective factor against SARI (OR: 0.07 95% CI [0.01–0.56]), household size (OR: 1.75, 95% CI [1.24–2.48]) and presence of secondhand smoke (OR: 2.2, 95% CI [1.12–4.45]) significant risk factors for SARI in infants. The mother alone being vaccinated is not a statistically significant protective factor against ILI (OR: 0.46, 95% CI [0.19–1.18]) or SARI (OR: 0.3, 95% CI [0.11–1.21]). Along with the obtained results and analysis, this study provides clear evidence that influenza vaccination of all household contacts of infants aged 0–6 months is significantly associated with protecting infants from both ILI and SARI.
Background
Influenza is a sudden viral illness triggered by an influenza virus infection. Classic symptoms of influenza commonly include sudden onset of high fever, upper respiratory symptoms like cough and nasal congestion, and muscle soreness. Severe outcomes of influenza include pneumonia, respiratory distress, encephalopathy, seizures, bloodstream infection, bacterial tracheitis, respiratory collapse, myocarditis, and mortality. Influenza may also develop as a co-infection with other respiratory viruses and may cause secondary bacterial infection.Citation1 Children account for roughly 13% of the worldwide influenza-infected population and face an increased likelihood of experiencing complications associated with influenza.Citation2 According to data collected by the Centers for Disease Control and Prevention’s (CDC) Influenza Hospitalization Surveillance Network spanning from 2010 to 2019, laboratory-confirmed influenza-related hospitalizations among children under 18 years old ranged from 10 to 375 cases per 100,000 individuals each season. Notably, infants under 6 months old exhibited the highest rates of hospitalization. Furthermore, the highest in-hospital mortality rates were observed in this same age group, with a rate of 0.73 deaths per 100,000 individuals.Citation3 The immunological immaturity of young children combined with their limited prior exposure to influenza increases their susceptibility to infection. As a consequence, there is often a notable increase in emergency room visits and hospitalizations due to influenza-related complications within this age cohort. Remarkably, severe influenza cases frequently manifest in children devoid of underlying health conditions. This vulnerability of children to influenza and its subsequent complications is a trend observed globally.Citation4
Given that vaccination stands as the most efficacious means of preventing influenza, both the Advisory Committee on Immunization Practices (ACIP) and the World Health Organization (WHO) advocate for influenza immunization of infants older than 6 months of age. Furthermore, they underscore the importance of prioritizing vaccination for young children owing to their heightened susceptibility to influenza-related complications.Citation5 During the 2017–2018 season, the overall vaccine effectiveness (VE) against both influenza A and B viruses was estimated at 38%. Notably, a higher VE of 64% was observed among children aged 6 months to 8 years in comparison to those aged 9 to 17 years, whose VE was 28%. In a separate study, the overall VE was reported as 65%, 25%, and 48% against influenza A (H1N1), A (H3N2), and B (B/Yamagata predominant), respectively.Citation6 In a systematic review and meta-analysis encompassing test-negative design studies focusing on influenza vaccine VE against laboratory-confirmed influenza-related hospitalizations in children aged 6 months to 17 years, researchers revealed significant findings. They determined that VE against influenza-associated hospitalizations in children aged 6 months to 5 years was 61.71% (95% CI: 49.29–74.12). Additionally, the study highlighted VE against hospitalization at 54.40% (95% CI: 33.37–75.43) in children aged 6 months to 2 years old, and notably higher at 73.14% (95% CI: 67.53–78.76) in children aged 2 to 5 years old.Citation7 While influenza-associated hospitalization rates were notably elevated among infants under 6 months old, it is crucial to note that current flu vaccine formulations are not authorized for administration in infants within their initial 6 months of life.Citation6 Offering passive protection to infants through maternal vaccination during pregnancy presents an attractive alternative to directly vaccinating infants.Citation8 Vaccinating pregnant and breastfeeding women results in a reduction in influenza incidence among infants. This is attributed to the transfer of maternal antibodies against influenza to the fetus through the placenta and to the infant through human milk. Studies have shown that levels of anti-influenza IgA in human milk remain significantly higher in mothers who receive the influenza vaccine during pregnancy for at least 6 months after delivery compared to non-vaccinated mothers.Citation9 Several studies have documented that seasonal influenza vaccination during pregnancy could provide significantly higher antibody levels in neonates and young infants than in those born to unvaccinated mothers.Citation10 Benowitz et al. showed that vaccination during pregnancy is effective in preventing hospitalization due to influenza in children in their first 12 months.Citation11 Evidence from numerous trials substantiates the safety of maternal influenza immunization for both pregnant women and infants, demonstrating no heightened risk of small-for-gestational-age birth, preterm birth, low birth weight, or fetal death.Citation12 Administering influenza vaccination during pregnancy presents a prime chance to avert influenza within the initial months of infancy. Moreover, ample evidence supports the safety of vaccine administration during pregnancy, leading to the adoption of policies in many countries recommending vaccination for pregnant women. Despite these guidelines, immunization rates among pregnant women persist below the desired thresholds across various settings, with less than 60% of women indicating that they received an influenza vaccine during pregnancy.Citation12,Citation13
An alternative and effective way to protect infants younger than 6 months from influenza is the practice of cocooning.Citation14 The term “cocoon vaccination strategy” denotes the practice of vaccinating individuals in the close vicinity of those who are susceptible to illness but unable to receive vaccinations due to contraindications or being too young for vaccination.Citation15 Cocooning may also decrease transmission. Bustamante et al. demonstrated that having a vaccinated grandparent serves as a protective factor. Adults, particularly parents of infants, have been identified as one of the primary sources of influenza transmission to young infants. Thus, vaccination of every member of the household in contact with the infant can be considered as an alternative to vaccination during pregnancy.Citation16 Since administering the current flu vaccine formulations to infants under 6 months old is not possible, we cannot directly measure the efficacy of the vaccine in children under 6 months of age. We conducted this study to assess the effectiveness of influenza vaccination among mothers and household contacts in reducing the occurrence of influenza-like illness (ILI) and severe acute respiratory infection (SARI) in infants. We also aimed to show whether interventions that emphasize parental education strategies affect acceptance of vaccination among household contacts of infants.
Methods
Study design
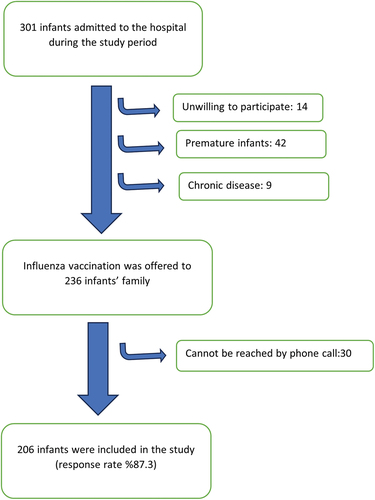
This prospective study took place in the well-child departments of Children’s Hospital in Turkey throughout the 2017–2018 influenza season and involved caregivers of infants aged 0–3 months. The hospital is a training and research center located in capital of Turkey, Ankara, and welcomes people from many different socioeconomic classes and provides free health services. Utilization of healthcare services is defined as both primary healthcare and hospital-based public visit. In Turkey, patients may apply to a tertiary hospital without a referral; thus, many children needing primary healthcare are seen at our hospital. Mothers whose neonates were seen in the social pediatric clinic for well-child visit in our hospital from October 1, 2017 to January 1, 2018 were considered to be eligible for study participation. A total of 1050 0–5-year-old children applied for well-child follow-up visits within 3 months; 301 of these children were between 0 and 3 months old. Parents/guardians who were unwilling to participate in the study, premature infants, and those with chronic diseases were excluded. The families of 236 infants who met the inclusion criteria were included in the study. The sample selection algorithm is shown in . This specific group was chosen as the target population for a possible future influenza cocooning vaccination program. Caregivers, parents, siblings, and grandparents of the infants younger than 3 months were included in the study.
In the first stage of the study, parents were invited and encouraged to participate in the study when they brought their baby for the first well-child visit (0–3 month). Parents completed informed consent and provided baseline measures of demographics and vaccination status. Participants were asked to complete a cross-sectional survey aimed at gathering fundamental demographic details concerning the infants and their families (e.g., age, sex, household composition, education status of the parents, influenza vaccination status of parents, cigarette usage, breastfeeding status). During the well-child visit, the clinician conducted healthy baby assessments as their usual standard of care. At the same time, the clinician provided information about influenza disease and safety, efficacy, and recommendation for a trivalent influenza vaccine (TIV). Following consultation with clinicians and screening for any contraindication for influenza vaccination (such as a history of severe allergic reaction after previous dose of influenza vaccine, history of Guillain-Barré syndrome within 6 weeks after receipt of influenza vaccine, or ongoing moderate or severe acute illness during the interview), mothers, fathers, and all household members of the neonates were presented with the opportunity to receive influenza vaccination. Influenza vaccination was administered upon voluntary acceptance of the participants. We divided the infants into three groups. The first group consisted of infants with no household contacts receiving an influenza vaccination (Group 1). The second group consisted of infants with a mother who is vaccinated but other members of the family are not (Group 2). The third group consisted of infants with all-household contacts vaccinated (Group 3). In the case in which the mother is vaccinated but other household members have mixed vaccination status, the cocoon was considered violated, and infants in this status were excluded from all three groups in order to avoid any bias and irregularity on the vaccine efficacy analysis.
Doctors contacted mothers by telephone every month during the influenza season (through the end of the June). The duration of follow-up after vaccination was between 6 and 9 months. During the telephone contact, mothers were asked about the number of people that live in the same house with the neonates/infants, whether or not they are vaccinated against influenza, and which household contacts had been vaccinated against influenza, if any. Medical history of the infants was elicited specifically for presence of the following symptoms within the previous 30 days from all caregivers of infants using standardized questionnaires: fever and/or respiratory symptoms (e.g., cough, runny nose, difficulty breathing), hospitalization, antibiotic usage, bronchodilator usage, and oxygen therapy. Participants were told to contact the researcher if their infants developed any symptoms between phone calls. Open-ended questions were used to collect information about reasons for vaccination and non-vaccination.
According to the severity of respiratory symptoms such as runny nose, cough, wheezing, or breathing difficulties, fever, required bronchodilator usage, oxygen therapy, antibiotic usage and hospitalization, we classified the participants as ILI, SARI, common cold, and asymptomatic. Clinical case definition of common cold is acute onset of runny nose, sneezing, nasal obstruction, and/or cough, without fever.Citation17 No treatment was given to these patients. The clinical case definition of influenza-like illness (ILI) encompasses an acute respiratory illness characterized by a recorded temperature of equal to or greater than 38°C along with cough, with symptom onset occurring within the preceding 10 days. The case definition of severe acute respiratory infection (SARI) involves an acute respiratory infection with either a history of fever or a measured fever of equal to or greater than 38°C, accompanied by cough, with symptoms emerging within the last 10 days, necessitating hospitalization.Citation18 Details of the illness definitions are shown in .
Table 1. Definition of illness.
Statistical analysis
Categorical and continuous variables were studied using the χ2 test and t-test, respectively. Following this, multivariate logistic regression analysis was employed to explore the association of independent variables in our dataset such as household size, maternal, paternal, and household vaccination status against influenza, passive smoking and breastfeeding status with our observations concerning ILI and SARI in infants. The effectiveness of influenza vaccine was calculated using the following formula: [1 – (incidence rate of ILI and SARI in infants with vaccinated only mothers and all household contacts/incidence rate of ILI and SARI in infants no household contacts receiving influenza vaccination) × 100]. Statistical analysis was conducted using SPSS 20 software tool.
Ethical approval
The study protocol was approved by local Ethics Committee of Sami Ulus Hospital and was conducted in adherence to the principles outlined in the Declaration of Helsinki (2017-141/375.).
Results
During the study period, influenza vaccination was offered to the families of 236 infants. The response rate was 87.3% (n:206). None of the mothers were vaccinated during pregnancy. The number of infants with at least one family member vaccinated was 64 (31.1%). Vaccination rate was 31.1% (n:64) among mothers and 23.3% (n:48) among fathers. 12.6% (n:26) of infants had only a mother who was vaccinated among all household contacts. The percent of infants for whom all household contacts were vaccinated against influenza was 16% (n:33). The percent of infants with none of the members of the family vaccinated was 68.9% (n:142). Five infants had a mother and one or more household contacts who were vaccinated but not all household contacts vaccinated, and thus they were excluded from data analysis.
The mean age of the mothers was 25.4 years (range 19–41 years). 77.6% of the parents had ≤ 2 children. 34.9% of the infants were exposed to tobacco smoke through their parents. 72.8% of the infants were exclusively breastfed. In terms of sociodemographic data, no statistically significant difference was detected between the groups in which mothers were vaccinated and in which they were not. When we compared the three groups, there were no differences in terms of mother’s education status (p = .08), father’s education status (p = .83), household size (p = .11), and breastfeeding status (p = .23). Only passive smoking rates were statically higher in infants with no members of the family vaccinated than the other groups (p < .05). Summary of infant characteristics is shown in .
Table 2. Distribution of Socio-demographic data according to infants’ maternal or household vaccination status.
Among the 206 infants enrolled in the study, over the six-month follow-up period, 35.9% (n:74) remained asymptomatic while 18.5% (n:38) experienced at least one episode of the common cold, 14.1% contracted influenza-like illness (ILI), and 31.5% were diagnosed with severe acute respiratory infection (SARI). 82.3% of those with SARI received antibiotic treatment. Illness distribution according to the maternal or household vaccination status is shown in . ILI and SARI rates among the infants with only the mother vaccinated (but other members of the family were not) were 34.6% and 23.1%, respectively. For fully cocooned infants, ILI and SARI rates were 27.3% and 9.1%, respectively. Among the infants whose mother was vaccinated (but other members of the family were not), the effectiveness of influenza vaccine was estimated as 35.3% for ILI and 41.3% for SARI. Among the infants whose parents and all people living with them were vaccinated against influenza, the effectiveness of influenza vaccine was estimated as 48.9% for ILI and 76.9% for SARI. The effectiveness of the influenza vaccine according to vaccination status of infants’ household contacts is shown in .
Table 3. Distribution of illness type according to infants’ maternal or household vaccination status.
Table 4. The effectiveness of the influenza vaccine according to vaccination status of infants’ household contacts.
Infants with no members of the family vaccinated had the highest risk for both ILI [OR = 2.05, 95% CI:1.15–3.56] and SARI [OR = 2.32, 95% CI:1.17–4.59]. The group with the lowest risk for ILI [OR = 0.36, 95% CI:0.16–0.83] and for SARI [OR = 0.17, 95% CI:0.05–0.58] was infants with all household contacts vaccinated. Vaccination of only mothers was not a statistically significant protective factor for ILI (OR: 0.46 95% CI [0.19–1.18]) or for SARI (OR: 0.3, 95% CI [0.11–1.21]). Risk estimation for ILI and SARI according to vaccination status of infants’ household contacts is shown in .
Table 5. Risk estimates for ILI and SARI according to vaccination status of infants’ household contacts.
Based on the results of multivariate logistic regression analysis, vaccination of all household members is a protective factor against SARI (OR: 0.07 95% CI [0.01–0.56]). Household size (OR: 1.75, 95% CI [1.24–2.48]) and presence of secondhand smoke (OR:2.2, 95% CI [1.12–4.45]) are significant risk factors for risk of SARI in infants ().
Table 6. Factors associated with severe acute respiratory infections.
For 93.7% (n:60) of the vaccinated mothers, the motivation to get vaccinated was to “protect their children” and for 0.6% (n:4) of the vaccinated mothers, their motivation was to protect themselves against influenza due to their existing chronic disease. In the non-vaccinated group, 57.1% (n:81) of the mothers thought that “influenza vaccination is unnecessary,” 36.6% (n:52) thought that “influenza vaccine is ineffective,” and 6.3% (n:9) had fears about its potential for causing acute adverse effects.
Discussion
Cocooning is immunizing everyone having close contact with a newborn or young infant against influenza. In this way, it is supposed that the cocoon, free from the danger of influenza, surrounds the infant until they can develop immunity and be eligible for vaccination at around 6 months old. Conducted in a tertiary children’s hospital, this study evaluated the acceptance and effectiveness of a cocooning strategy against influenza. Our results show that after recommendation, the vaccination rate was 31.1% among mothers and 23.3% among fathers, and for 16% of babies, all household contacts were vaccinated (fully cocooned). The effectiveness of influenza vaccine among infants with only mothers vaccinated is estimated as 35.3% for ILI and 41.3% for SARI. Among fully cocooned infants, the effectiveness of influenza vaccine is 48.9% for ILI and 76.9% for SARI. Infants with no members of the family vaccinated had highest risk for both ILI [OR = 2.05, 95% CI:1.15–3.56] and SARI [OR = 2.32, 95% CI:1.17–4.59]. Based on the results of multivariate logistic regression analysis, vaccination of all household members is a protective factor for both ILI and SARI, and vaccination of only the mother is not a statistically significant protective factor against ILI and SARI. In other words, vaccination of only mothers is not sufficient for effectively protecting infants. Postpartum maternal vaccination works under the condition that a cocoon is established around the infant by vaccinating all household members. While studies on the effectiveness of maternal influenza vaccination are at the forefront in the literature, the number of studies on the effect of vaccinating the entire household is very limited. K Zaman et al. demonstrated that maternal immunization with influenza vaccine exhibited notable clinical efficacy, resulting in a substantial reduction of 63% in laboratory-confirmed influenza illness and a 29% reduction in rates of respiratory illness with fever among infants up to 6 months old.Citation19 Andrea G et al. reported that in Mali, households where both mothers and other household contacts of infants received the influenza vaccine experienced fewer cases of laboratory-confirmed influenza (LCI). Additionally, there was a discernible trend indicating reduced rates of H1N1-LCI within these households.Citation20 A similar study conducted in Greece revealed that 73.7% of the infants’ mothers were vaccinated. The authors analyzed vaccine effectiveness in 530 infants, revealing that postpartum maternal vaccination exhibited 53.5% effectiveness against ILI and 45.4% effectiveness against the administration of antibiotics. These results are in line with a study conducted by Maltezeu et al., demonstrating consistency in findings across studies.Citation21 In addition to the effect of mothers’ vaccination, which was studied in Maltezeu’s work, we also investigated the effects of vaccination of household individuals who constitute a cocoon. Accordingly, we observed that the frequency of ILI and SARI in fully cocooned babies decreased significantly compared to other groups. Another study investigating the impact of maternal influenza vaccination on influenza virus infection in young infants revealed that the incidence rate of ILI was 7.2 and 6.7 per 1000 person-days for infants born to unvaccinated and vaccinated women, respectively. Significantly, infants born to mothers vaccinated against influenza demonstrated a 41% decrease in the risk of laboratory-confirmed influenza virus infection and a 39% decrease in the risk of hospitalization due to influenza-like illness (ILI) compared to infants born to unvaccinated mothers. However, it is important to note that this study solely assessed the effect of seasonal influenza vaccination during pregnancy.Citation22
Based on the results of multivariate logistic regression analysis, whole household vaccination is a protective factor against both ILI and SARI. Only the mother being vaccinated is not a statistically significant protective factor against SARI. The group with the highest vaccine effectiveness is the group of infants for whom all-household contacts were vaccinated against influenza. The effectiveness of cocooning as a strategy to achieve lasting control of infections hinges upon several key factors, including healthcare provider advocacy, patient education, the implementation and enforcement of policies, and the development of cost-effective programs.Citation23 The mother’s status of already being vaccinated against influenza is the main factor that increases the acceptance rate of the cocoon by the household.Citation24 In our study, we observed that if there are vaccinated individuals in the family, mothers are always one of the vaccinated individuals, which means that mothers are much more motivated about protection of their babies. Although we do not know whether the influenza vaccine, which is routinely recommended during pregnancy, was previously recommended by a healthcare professional to the mothers participating in this study, we found that none of the mothers received the influenza vaccine during their pregnancies. In this case, the second option, cocooning strategy to protect infants, was recommended to families in this study, and their vaccination status was checked during follow-up. We observed that all household contacts of 16% of the children were vaccinated. In order to increase this rate, it might be more effective to give and implement the cocoon recommendation under hospital conditions as soon as the baby is born. In 2009, New York State enacted the New York State Neonatal Influenza Protection Act (NIPA), mandating hospitals providing newborn services to offer influenza vaccination to caregivers of newborns. Following the implementation of the NIPA vaccination policy, a notable decrease in the proportion of total infant influenza cases was observed. The study indicated that the overall effectiveness of influenza vaccines during the study period ranged from 50% to 70%. In the NIPA study, the effectiveness of the vaccine was solely assessed in children whose parents received vaccination; however, it is crucial to note that parental vaccination alone may not suffice to establish cocoon immunity.Citation25 In our study, vaccine effectiveness was examined in those with a full cocoon and was found to be 48.9% for ILI and 76.9% for SARI. Although the cocoon strategy seems to be an expensive practice, it can actually be shown to be cost effective with a cost-effectiveness study considering all variables, as it protects infants from antibiotic usage and hospitalization. Bustamante et al. showed that having at least one vaccinated grandparent was an independent protective factor (OR 0.22 [95% CI: 0.05–0.91]) against hospital admission due to influenza.Citation16 Similarly, in our study we found that being in full cocoon was an independent protective factor against ILI [OR = 0.36, 95% CI:0.16–0.83] and against SARI [OR = 0.17, 95% CI:0.05–0.58].
We found that household size and presence of secondhand smoke were greatest risk factors for SARI in infants. Influenza is a highly contagious airway infection that spreads easily among individuals in close proximity through large droplets produced during coughing or sneezing as well as through direct contact. In a study conducted by Pavani K et al., children residing in crowded households experienced a 60% increase in the odds of pneumonia.Citation26 In a separate study by Oguz et al., a notable correlation was found between the presence of at least one reported respiratory symptom within the past week and the size of the household.Citation27 Conversely, Joshua G et al. did not find any correlation between household size or congestion, household vaccination coverage, humidification, or exposure to tobacco smoke and the probability of influenza introduction.Citation28 In our study of 206 infants, 35% were exposed to secondhand smoke (SHS). The SHS exposure rate was higher in infants with no members of the family vaccinated than the other groups. Children with SHS exposure were determined to be 2.2 times more likely to have SARI. However, data about the association with SHS exposure and potential mechanisms of viral infections are limited. In this regard, in a study on an adult population, cigarette smoking was found to be associated with higher mortality in patients with influenza.Citation29,Citation30 It was shown that children with confirmed influenza and secondhand tobacco smoke exposure were more likely to require intensive care and had a longer length of stay in hospital than children not exposed to secondhand tobacco smoke.Citation30 Another study about the pathophysiological link between viral illness and tobacco smoke exposure suggests that the suppression of interferon-ɣ and concurrent activation of Th2 responses may contribute, at least partially, to increased severity of illness.Citation31
A study in Turkey was designed to determine the attitude of pregnant women about immunizations during pregnancy. Among 786 pregnant participants, only 1% of them had seasonal influenza vaccine after the healthcare provider gave information about influenza vaccination.Citation32 In our study, the vaccination rate against influenza during pregnancy was 0%. After we gave information about influenza and cocooning, 31.1% of the mothers were vaccinated postdelivery. Studies conducted in various countries where influenza immunization is recommended for pregnant women have reported immunization rates ranging from 20% to 66%.Citation33 Despite numerous organizations advocating for cocooning vaccination of household contacts of infants under 12 months old against influenza, widespread implementation at a national level has been impeded by logistical and financial obstacles.Citation34 On the contrary, despite the Turkish Ministry of Health providing influenza vaccine to pregnant women free of charge, the vaccination rate against influenza during pregnancy remains notably low. In a cross-sectional survey conducted by N. Yakut et al. involving 465 pregnant women in Turkey, only 19.8% of participants accepted influenza vaccination when offered.Citation35 Similarly, in another study by Celikel et al., it was noted that merely 3% of pregnant women accepted influenza vaccination during pregnancy.Citation36 A meta-analysis examining factors influencing vaccination decision-making among pregnant women highlighted prejudice against vaccination as one of the most significant reasons for the poor vaccination rates.Citation37 Although the Ministry of Health routinely recommends flu vaccination for pregnant women, none of the pregnant women included in our study had been vaccinated during pregnancy. Since we did not ask about whether vaccination was recommended to the participant mothers during pregnancy in our study, we did not have the opportunity to compare the effect of the vaccination recommendation given by us in the postnatal follow-up well-child visits. Based on our findings, it is evident that counseling regarding influenza and cocooning during well-child visits significantly boosts vaccine acceptance. Saglam et al. revealed that approximately half of the pregnant women (46.0%) were uninformed about the existence of an influenza vaccine.Citation38 This rate also shows that pregnant women are not given adequate information about influenza vaccination. Previous studies conducted across various demographic groups have consistently indicated that the recommendations of healthcare workers can significantly enhance the rate of vaccination coverage during pregnancy.Citation39,Citation40 We show that the vaccination rates of mothers may be increased by informing them about influenza and cocooning. As evidenced by prior studies, the recommendation from healthcare providers emerges as a paramount intervention in enhancing maternal vaccination rates.Citation41 It underscores the significance of delivering vaccine counseling consistently throughout pregnancy and during well-child visits.
In our study, 68.9% of the infants had no family members vaccinated. When we asked only mothers why they were not vaccinated, the most common reasons were “influenza vaccination is unnecessary,” “influenza vaccine is ineffective,” and fears about its potential for causing acute adverse effects. Previous studies reported that the side effects of the vaccine and efficacy concerns were the most common reasons for vaccination rejection.Citation42 Terzi et al. demonstrated that the most prevalent reasons for vaccine refusal among parents were “fear of vaccine side-effects” and “issues related to previous vaccinations.”Citation43 A study conducted in Turkey regarding influenza vaccine acceptance among parents found that the most significantly associated factors for vaccine refusal were the belief that the influenza pandemic was exaggerated and the perception that other preventive measures were more effective than vaccination.Citation44 In another study investigating childhood vaccine refusal rates and causes in Turkey during 2016–2017, the refusal rate was observed to be 5.9%. This study highlighted concerns about the vaccine content, perceived harmfulness, and fears about adverse effects as the most common reasons underlying vaccine refusal.Citation45
The largest number of studies focused on the influenza vaccine were due to the 2009 Influenza A (H1N1) pandemic, and most were published in 2011. Studies on influenza vaccination have relatively lost their importance with the COVID-19 pandemic. However, with the removal of COVID-19 pandemic measures, an increase in the frequency of influenza has been observed.Citation46 This serves as a reminder of the critical importance of influenza vaccination. Regrettably, children remain vulnerable to influenza exposure and are at increased risk of complications from the virus. While annual influenza vaccination is recommended for all children, vaccination rates among healthy children remain suboptimal. As the findings of our study imply, it is noteworthy that well-child visits present an opportune moment to recommend cocooning strategies during the influenza season.
In the absence of laboratory confirmation of influenza diagnosis, several clinical case definitions have been used for influenza surveillance. These include influenza-like illness (ILI), severe acute respiratory infection (SARI), acute respiratory infection (ARI), and febrile acute respiratory illness (FARI). In our study, instead of laboratory-confirmed influenza virus infection, we only used definitions based on reported symptoms. This is the main limitation of our study. Since influenza-related diseases can present with very different clinical findings, the use of standard definitions may cause the disease to be missed. There are many studies aiming to increasing the sensitivity and specificity of identification.Citation47–49 We used the World Health Organization’s (WHO) case definitions for both ILI and SARI, which were developed in 2011 for influenza surveillance. They are based on a combination of signs and symptoms. During peak influenza season, other respiratory viruses also peak, so this may increase the possibility of misdiagnosing cases with influenza. To reduce the misidentification of influenza, we can use a higher temperature cutoff for fever and/or include rhinorrhea or other additional respiratory symptoms to increase specificity. Shital C et al. showed that the best predictive model for identifying influenza for all ages consisted of cough, fever, rhinorrhea, and myalgias.Citation49,Citation50 We defined ILI, as does WHO, as acute respiratory illness with a measured temperature of ≥ 38°C and cough with onset within the past 10 days. If we had included the presence of fever over ≥ 39°C along with cough in the definition of ILI, we could have increased the specificity further, but in this case, the sensitivity would have decreased. Vivek et al., in their clinical case definition validation study for influenza in 1043 hospitalized patients, 257 of whom were children, found the sensitivity for measured ⁄ reported fever plus cough to be 74% and specificity to be 61% for the detection of ILI. They found that if the case definitions included only measured fever, then sensitivity would be lower.Citation51
Choosing monthly follow-ups over weekly ones may have resulted in missed occurrences of SARI or ILI episodes in between, thereby potentially influencing the outcomes. This limitation should be taken into consideration. Calling families at shorter intervals would undoubtedly have yielded results that are more reliable. However, it would have been more costly and much more laborious for both the researcher and the participants. Instead, along with the monthly calls, participants were told to contact the researcher if their infants developed any symptoms between phone calls. Relying solely on patient self-reporting is inherently unreliable, as some patients may provide accurate information while others may not. Therefore, this aspect represented another limitation of our study. This study did not include infants aged 3–6 months because they would have become eligible for vaccination during the influenza season, thus potentially confounding the results.
Our study provides clear evidence that influenza vaccination of all household contacts of infants is significantly associated with protecting infants from ILI and SARI during the first 6 months of life. The frequencies of ILI and SARI were lowest in the group in which all household contacts were vaccinated. Our findings strongly support the recommendations for the cocooning strategy. In order to increase the use of influenza vaccines for pregnant women, information about influenza and vaccination should be provided by health professionals. Appropriate policies can be developed for pregnancy vaccination and cocoon strategy implementation by designing studies that better understand the attitudes of pregnant women, other caregivers, and society toward vaccination. For those who refuse vaccination during pregnancy, the woman could also be advised to consider the cocoon strategy at her prenatal visits, which would give more time for other household contacts to decide to be vaccinated. Additionally, future studies with laboratory-confirmed influenza cases should be planned to investigate the effectiveness of the cocoon strategy for more reliable results.
Author contribution
Oguz MM and Senel S and designed the study; Oguz MM collected and analyzed data; Oguz MM wrote the manuscript. Senel S and Oguz MM made important intellectual/conceptual critical corrections for preparation of study design and for the content. All authors read and approved the final manuscript. The authors report there are no competing interests to declare.
Disclosure statement
No potential conflict of interest was reported by the author(s).
Additional information
Funding
References
- Mistry RD, Fischer JB, Prasad PA, Coffin SE, Alpern ER. Severe complications in influenza-like illnesses. Pediatrics. 2014;134(3):684–10. doi:10.1542/peds.2014-0505.
- Ruf BR, Knuf MJ. The burden of seasonal and pandemic influenza in infants and children. Eur J Pediatr. 2014;173(3):265–76. doi:10.1007/s00431-013-2023-6.
- Kamidani S, Garg S, Rolfes MA, Campbell AP, Cummings CN, Haston JC, Openo KP, Fawcett E, Chai SJ, Herlihy R., et al. Epidemiology, clinical characteristics, and outcomes of influenza-associated hospitalizations in US children over 9 seasons following the 2009 H1N1 pandemic. Clin Infect Dis. 2022;75(11):1930–9. doi:10.1093/cid/ciac296.
- Fisher WA, Gilca V, Murti M, Orth A, Garfield H, Roumeliotis P, Rampakakis E, Brown V, Yaremko J, Van Buynder P., et al. Parental attitudes and perceptions of support after brief clinician intervention predict intentions to accept the adjuvanted seasonal influenza vaccination: findings from the Pediatric Influenza Vaccination Optimization Trial (PIVOT)–I. Vaccines. 2022;10(11):1957. doi:10.3390/vaccines10111957.
- Fisher WA, Gilca V, Murti M, Orth A, Garfield H, Roumeliotis P, Rampakakis E, Brown V, Yaremko J, Van Buynder P., et al. Clinicians are not able to infer parental intentions to vaccinate infants with a seasonal influenza vaccine, and perhaps they should not try: findings from the Pediatric Influenza Vaccination Optimization Trial (PIVOT)–IV. Vaccines. 2022;10(11):1955. doi:10.3390/vaccines10111955.
- Maldonado YA, Zaoutis TE, Banerjee R, Barnett ED, Campbell JD, Caserta MT, Gerber JS, Kourtis AP, Lynfield R, Nolt D., et al. Recommendations for prevention and control of influenza in children, 2019–2020. Pediatrics. 2019;144(4). doi:10.1542/peds.2019-2478.
- Kalligeros M, Shehadeh F, Mylona EK, Dapaah-Afriyie C, van Aalst R, Chit A, Mylonakis E. Influenza vaccine effectiveness against influenza-associated hospitalization in children: a systematic review and meta-analysis. Vaccine. 2020;38(14):2893–903. doi:10.1016/j.vaccine.2020.02.049.
- Nunes MC, Madhi SAJ. Influenza vaccination during pregnancy for prevention of influenza confirmed illness in the infants: a systematic review and meta-analysis. Hum Vaccines Immunother. 2018;14(3):758–66. doi:10.1080/21645515.2017.1345385.
- Hunagund S, Golan Y, Asiodu IV, Prahl M, Gaw SLJ. Effects of vaccination against influenza, pertussis, and COVID-19 on human milk antibodies: current evidence and implications for health equity. Front Immunol. 2022;13:910383. doi:10.3389/fimmu.2022.910383.
- Principi N, Senatore L, Esposito SJ. Protection of young children from influenza through universal vaccination. Hum Vaccines Immunother. 2015;11(10):2350–8. doi:10.1080/21645515.2015.1055428.
- Benowitz I, Esposito DB, Gracey KD, Shapiro ED, Vázquez MJ. Influenza vaccine given to pregnant women reduces hospitalization due to influenza in their infants. Clin Infect Dis. 2010;51(12):1355–61. doi:10.1086/657309.
- Regan AK, Munoz FM. Efficacy and safety of influenza vaccination during pregnancy: realizing the potential of maternal influenza immunization. Expert Rev Vaccines. 2021;20(6):649–60. doi:10.1080/14760584.2021.1915138.
- Razzaghi H, Kahn KE, Black CL, Lindley MC, Jatlaoui TC, Fiebelkorn AP, Havers FP, D’Angelo DV, Cheung A, Ruther NA., et al. Influenza and Tdap vaccination coverage among pregnant women—United States, April 2020. MMWR Morb Mortal Wkly Rep. 2020;69(39):1391–7. doi:10.15585/mmwr.mm6939a2.
- Pediatrics CoIDJ. Recommendations for prevention and control of influenza in children, 2021–2022. Pediatrics. 2021;148(4). doi:10.1542/peds.2021-053744.
- Nitsch-Osuch A. Cocoon strategy of vaccinations: benefits and limitations. IntechOpen; 2017.
- Bustamante J, Calzado I, Sainz T, Calvo C, Del Rosal T, Méndez-Echevarría AJ. Epidemiological factors related to hospitalization due to influenza in children below 6 months of age. Eur J Pediatr. 2017;176(10):1425–8. doi:10.1007/s00431-017-3001-1.
- Heikkinen T, Järvinen AJTL. The common cold. Lancet. 2003;361(9351):51–9. doi:10.1016/S0140-6736(03)12162-9.
- Fitzner J, Qasmieh S, Mounts AW, Alexander B, Besselaar T, Briand S, Brown C, Clark S, Dueger E, Gross D., et al. Revision of clinical case definitions: influenza-like illness and severe acute respiratory infection. Bull World Health Organ. 2018;96(2):122. doi:10.2471/BLT.17.194514.
- Zaman K, Roy E, Arifeen SE, Rahman M, Raqib R, Wilson E, Omer SB, Shahid NS, Breiman RF, Steinhoff MC., et al. Effectiveness of maternal influenza immunization in mothers and infants. N Engl J Med. 2008;359(15):1555–64. doi:10.1056/NEJMoa0708630.
- Buchwald AG, Tamboura B, Haidara FC, Coulibaly F, Doumbia M, Diallo F, Boudova S, Keita AM, Sow SO, Kotloff K., et al. Maternal influenza vaccination and the risk of laboratory-confirmed influenza among household contacts under the age of five in Mali. Am J Trop Med Hyg. 2019;100(1):159. doi:10.4269/ajtmh.18-0450.
- Maltezou HC, Fotiou A, Antonakopoulos N, Kallogriopoulou C, Katerelos P, Dimopoulou A, Tsoutsa V, Siahanidou T, Papagaroufalis C, Kostis E., et al. Impact of postpartum influenza vaccination of mothers and household contacts in preventing febrile episodes, influenza-like illness, healthcare seeking, and administration of antibiotics in young infants during the 2012–2013 influenza season. Clin Infect Dis. 2013;57(11):1520–6. doi:10.1093/cid/cit599.
- Eick AA, Uyeki TM, Klimov A, Hall H, Reid R, Santosham M, O’Brien KL. Maternal influenza vaccination and effect on influenza virus infection in young infants. Arch Pediatr Adolesc Med. 2011;165(2):104–11. doi:10.1001/archpediatrics.2010.192.
- Grizas AP, Camenga D, Vázquez MJ. Cocooning: a concept to protect young children from infectious diseases. Curr Opin Pediatr. 2012;24(1):92–7. doi:10.1097/MOP.0b013e32834e8fe9.
- O’Leary ST, Riley LE, Lindley MC, Allison MA, Albert AP, Fisher A, Jiles AJ, Crane LA, Hurley LP, Beaty B, et al. Obstetrician–gynecologists’ strategies to address vaccine refusal among pregnant women. Obstet Gynecol. 2019;133(1):40–7. doi:10.1097/AOG.0000000000003005.
- Shah S, Messina CJ. The time-associated impact of the newborn influenza protection act on infant influenza rates in New York State. J Perinat Med. 2014;42(6):711–6. doi:10.1515/jpm-2014-0248.
- Ram PK, Dutt D, Silk BJ, Doshi S, Rudra CB, Abedin J, Goswami D, Fry AM, Brooks WA, Luby SP, et al. Household air quality risk factors associated with childhood pneumonia in urban Dhaka, Bangladesh. Am J Trop Med Hyg. 2014;90(5):968. doi:10.4269/ajtmh.13-0532.
- Oguz M, Camurdan A, Aksakal F, Akcaboy M, Acoglu EA. Social contact patterns of infants in deciding vaccination strategy: a prospective, cross-sectional, single-centre study. Epidemiol Infect. 2018;146(9):1157–66. doi:10.1017/S0950268818001048.
- Petrie JG, Ohmit SE, Cowling BJ, Johnson E, Cross RT, Malosh RE, Thompson MG, Monto AS. Influenza transmission in a cohort of households with children: 2010–2011. PLOS ONE. 2013;8(9):e75339. doi:10.1371/journal.pone.0075339.
- Karadag‐Oncel E, Ciblak MA, Ozsurekci Y, Badur S, Ceyhan MJ. Viral etiology of influenza‐like illnesses during the influenza season between December 2011 and April 2012. J Med Virol. 2014;86(5):865–71. doi:10.1002/jmv.23747.
- Wilson KM, Pier JC, Wesgate SC, Cohen JM, Blumkin AKJ. Secondhand tobacco smoke exposure and severity of influenza in hospitalized children. J Pediatr. 2013;162(1):16–21. doi:10.1016/j.jpeds.2012.06.043.
- Marseglia GL, Avanzini MA, Caimmi S, Caimmi D, Marseglia A, Valsecchi C, Poddighe D, Ciprandi G, Pagella F, Klersy C., et al. Passive exposure to smoke results in defective interferon-γ production by adenoids in children with recurrent respiratory infections. J Interferon Cytokine Res. 2009;29(8):427–32. doi:10.1089/jir.2008.0108.
- Celep G, Çamurdan AD, Aksakal FNB, Kara OF. Different perspectives of immunizations during pregnancy. Turk J Med Sci. 2020;50(2):316–23. doi:10.3906/sag-1910-23.
- Erb ML, Erlanger T, Heininger UJVX. Child-parent immunization survey: how well are national immunization recommendations accepted by the target groups? Vaccine. 2019;1:100013. doi:10.1016/j.jvacx.2019.100013.
- Grohskopf LA. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—United States, 2019–20 influenza season. MMWR Recomm Rep. 2019;68(3):1–21.
- Yakut N, Soysal S, Soysal A, Bakir MJ. Knowledge and acceptance of influenza and pertussis vaccinations among pregnant women of low socioeconomic status in Turkey. Hum Vaccines Immunother. 2020;16(5):1101–8. doi:10.1080/21645515.2019.1689082.
- Celikel A, Ustunsoz A, Guvenc GJ. Determination of vaccination status of pregnant women during pregnancy and the affecting factors. J Clin Nurs. 2014;23(15–16):2142–50. doi:10.1111/jocn.12351.
- Kilich E, Dada S, Francis MR, Tazare J, Chico RM, Paterson P, Larson HJ. Factors that influence vaccination decision-making among pregnant women: a systematic review and meta-analysis. PLOS ONE. 2020;15(7):e0234827. doi:10.1371/journal.pone.0234827.
- Sağlam M, Öncel S, Gündoğdu Z, Öncel S, Gundogdu ZJC. Knowledge, attitude, perceptions, and concerns of pregnant women regarding the influenza vaccination in Kocaeli, Turkey. Cureus. 2022;14(4). doi:10.7759/cureus.23765.
- Krishnaswamy S, Cheng AC, Wallace EM, Buttery J, Giles MLJ. Understanding the barriers to uptake of antenatal vaccination by women from culturally and linguistically diverse backgrounds: a cross-sectional study. Hum Vaccines Immunother. 2018;14(7):1591–8. doi:10.1080/21645515.2018.1445455.
- Costantino C, Mazzucco W, Bonaccorso N, Cimino L, Conforto A, Sciortino M, Catalano G, D’Anna MR, Maiorana A, Venezia R., et al. Educational interventions on pregnancy vaccinations during childbirth classes improves vaccine coverages among pregnant women in Palermo’s province. Vaccines. 2021;9(12):1455. doi:10.3390/vaccines9121455.
- Bisset KA, Paterson PJV. Strategies for increasing uptake of vaccination in pregnancy in high-income countries: a systematic review. Vaccine. 2018;36(20):2751–9. doi:10.1016/j.vaccine.2018.04.013.
- Albattat HS, Alahmed AA, Alkadi FA, Aldrees OSJ. Knowledge, attitude, and barriers of seasonal influenza vaccination among pregnant women visiting primary healthcare centers in Al-Ahsa, Saudi Arabia. 2019/2020. J Family Med Prim Care. 2021;10(2):783. doi:10.4103/jfmpc.jfmpc_2183_20.
- Terzi Ö, Gulen E, Dündar CJ. The causes of parental vaccine refusal: results of a survey from Giresun, Turkey. Turk J Pediatr. 2021;63(4):618–25. doi:10.24953/turkjped.2021.04.009.
- Akıs S, Velıpasaoglu S, Camurdan AD, Beyazova U, Sahın FJ. Factors associated with parental acceptance and refusal of pandemic influenza A/H1N1 vaccine in Turkey. Eur J Pediatr. 2011;170(9):1165–72. doi:10.1007/s00431-011-1425-6.
- Yalçin SS, Kömürlüoğlu A, Topaç OJ. Rates of childhood vaccine refusal in Turkey during 2016–2017: regional causes and solutions. Archives de Pédiatrie. 2022;29(8):594–8. doi:10.1016/j.arcped.2022.06.005.
- Zeng H, Cai M, Li S, Chen X, Xu X, Xie W, Xiong Y, Long X. Epidemiological characteristics of seasonal influenza under implementation of zero-COVID-19 strategy in China. J Infect Public Health. 2023;16(8):1158–66. doi:10.1016/j.jiph.2023.05.014.
- Jiang L, Lee V, Lim W, Chen M, Chen Y, Tan L, Lin RT, Leo YS, Barr I, Cook AR., et al. Performance of case definitions for influenza surveillance. Eurosurveillance. 2015;20(22):21145. doi:10.2807/1560-7917.ES2015.20.22.21145.
- Domínguez À, Soldevila N, Torner N, Martínez A, Godoy P, Rius C, Jané M, PIDIRAC Sentinel Surveillance Program of Catalonia T. Usefulness of clinical definitions of influenza for public health surveillance purposes. Viruses. 2020;12(1):95. doi:10.3390/v12010095.
- Casalegno J-S, Eibach D, Valette M, Enouf V, Daviaud I, Behillil S, Vabret A, Soulary JC, Benchaib M, Cohen JM., et al. Performance of influenza case definitions for influenza community surveillance: based on the French influenza surveillance network GROG, 2009–2014. Eurosurveillance. 2017;22(14):30504. doi:10.2807/1560-7917.ES.2017.22.14.30504.
- Shah SC, Rumoro DP, Hallock MM, Trenholme GM, Gibbs GS, Silva JC, Waddell MJ. Clinical predictors for laboratory-confirmed influenza infections: exploring case definitions for influenza-like illness. Infect Cont Hosp Ep. 2015;36(3):241–8. doi:10.1017/ice.2014.64.
- Gupta V, Dawood FS, Rai SK, Broor S, Wigh R, Mishra AC, Lafond K, Mott JA, Widdowson M-A, Lal RB., et al. Validity of clinical case definitions for influenza surveillance among hospitalized patients: results from a rural community in North India. Influenza Other Respir Viruses. 2013;7(3):321–9. doi:10.1111/j.1750-2659.2012.00401.x.