Abstract
Background: A dual COX/5-LOX strategy was adopted to develop new oxindole derivatives with superior anti-inflammatory activity. Methods: Three series of oxindoles – esters 4a–p, 6a–l and imines 7a–o – were synthesized and evaluated for their anti-inflammatory and analgesic activities. Molecular docking and predicted pharmacokinetic parameters were done for the most active compounds. A new LC–MS/MS method was developed and validated for the quantification of 4h in rat plasma. Results: Compounds 4h, 6d, 6f, 6j and 7m revealed % edema inhibition up to 100.00%; also, 4l and 7j showed 100.00% writhing protection. Compound 4h showed dual inhibitory activity with IC50 = 0.0533 and 0.4195 μM for COX-2 and 5-LOX, respectively. Molecular docking rationalized the obtained biological activity. The pharmacokinetic parameters of 4h from rat plasma were obtained.
Graphical abstract
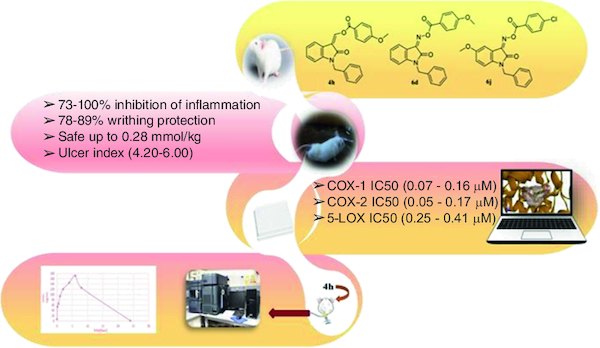
Different oxindole-based derivatives were synthesized.
The new compounds showed potent anti-inflammatory and analgesic activities.
Compounds 4h, 6d, 6f, 6j and 7m were the most active as anti-inflammatory.
Compounds 4l and 7g showed the best analgesic activity.
Compound 4h showed high COX-2 inhibitory activity with selectivity index of 3.07.
Compounds 6d and 6j revealed potent 5-LOX inhibitory activity.
The molecular docking studies confirmed the structure–activity relationship study and the enzyme inhibition results.
Promising pharmacokinetic parameters of compounds 4h, 6d and 6j were obtained using the free webserver Swiss Institute of Bioinformatics SwissADME®.
In vivo pharmacokinetic parameters of compound 4h were obtained from rat plasma using a newly developed MS/MS method.
Financial disclosure
The authors have no financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
Competing interests disclosure
The authors have no competing interests or relevant affiliations with any organization or entity with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
Writing disclosure
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations.

